Organic reaction
Claisen Rearrangement[edit]
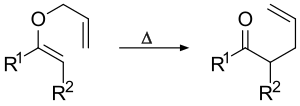
The Claisen rearrangement is a fundamental organic reaction that involves the [3,3]-sigmatropic rearrangement of an allyl vinyl ether to produce a _,_-unsaturated carbonyl compound. This reaction is named after the German chemist Ludwig Claisen, who first reported it in 1912.
Mechanism[edit]
The Claisen rearrangement is a concerted process, meaning that it occurs in a single step without the formation of intermediates. The reaction proceeds through a six-membered cyclic transition state, which is a hallmark of pericyclic reactions. The driving force for the rearrangement is the formation of a more stable carbonyl compound from the less stable allyl vinyl ether.
Variants[edit]
Several variants of the Claisen rearrangement exist, including:
- The Johnson–Claisen rearrangement, which involves the use of an orthoester to generate the allyl vinyl ether in situ.
- The Ireland–Claisen rearrangement, which uses a silyl ketene acetal as the starting material and is performed under basic conditions.
- The Eschenmoser–Claisen rearrangement, which involves the rearrangement of allylic amines.
Applications[edit]
The Claisen rearrangement is widely used in organic synthesis to form carbon-carbon bonds and to introduce functional groups into molecules. It is particularly useful in the synthesis of natural products and complex organic molecules.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian