Peroxide
Peroxide[edit]
A peroxide is a chemical compound that contains a peroxide group, which is characterized by an oxygen-oxygen single bond (O-O). The general formula for a peroxide is R-O-O-R, where R can be a hydrogen atom or an organic group. Peroxides are a subclass of reactive oxygen species and are known for their strong oxidizing properties.
Structure and Bonding[edit]
The peroxide group consists of a single bond between two oxygen atoms. This bond is relatively weak compared to other oxygen bonds, such as the double bond in oxygen gas (O₂). The O-O bond length in peroxides is typically around 1.49 Å, which is longer than the O=O bond length in oxygen gas, which is about 1.21 Å.
Types of Peroxides[edit]
Peroxides can be classified into several types based on their structure and the nature of the R groups:
Hydrogen Peroxide[edit]
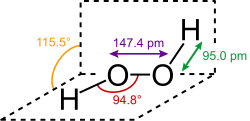
Hydrogen peroxide (H₂O₂) is the simplest and most well-known peroxide. It is a pale blue liquid in its pure form and is commonly used as a disinfectant and bleaching agent. Hydrogen peroxide decomposes into water and oxygen, a reaction that is catalyzed by light, heat, and various catalysts.
Organic Peroxides[edit]
Organic peroxides contain the peroxide group bonded to organic moieties. They are often used as initiators in polymerization reactions and as bleaching agents. Due to their instability, organic peroxides are often handled with care to prevent explosive decomposition.
Inorganic Peroxides[edit]
Inorganic peroxides include compounds such as sodium peroxide (Na₂O₂) and barium peroxide (BaO₂). These compounds are typically used in industrial applications, such as bleaching and as oxidizing agents.
Applications[edit]
Peroxides have a wide range of applications in various industries:
- Bleaching Agents: Peroxides are used in the bleaching of paper and textiles.
- Disinfectants: Hydrogen peroxide is used as a disinfectant in medical and household settings.
- Polymerization Initiators: Organic peroxides are used to initiate the polymerization of monomers in the production of plastics.
- Rocket Propellants: Some peroxides are used as oxidizers in rocket propellants.
Safety and Handling[edit]
Peroxides can be hazardous due to their reactive nature. They can decompose explosively under certain conditions, releasing oxygen gas and heat. Proper storage and handling procedures are essential to minimize the risk of accidents.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian