Β-Lactam antibiotic
Class of broad-spectrum antibiotics
β-Lactam antibiotics are a class of broad-spectrum antibiotics that contain a β-lactam ring in their molecular structures. This class includes penicillins, cephalosporins, carbapenems, and monobactams, which are among the most widely used antibiotics in the world.
Structure and Mechanism of Action[edit]
The β-lactam ring is a four-membered lactam, which is a cyclic amide. The integrity of this ring is crucial for the antibiotic activity of these compounds. β-Lactam antibiotics work by inhibiting the synthesis of the bacterial cell wall, specifically by targeting the penicillin-binding proteins (PBPs) that are essential for cell wall construction.
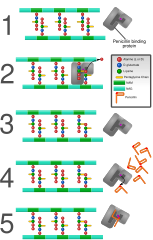
The inhibition of PBPs prevents the cross-linking of peptidoglycan chains, which is a critical step in cell wall biosynthesis. This leads to the weakening of the cell wall and eventually causes cell lysis and death, particularly in actively dividing bacteria.
Types of β-Lactam Antibiotics[edit]
Penicillins[edit]
Penicillins were the first β-lactam antibiotics discovered and are still widely used today. They are effective against a variety of Gram-positive and some Gram-negative bacteria. Common penicillins include penicillin G, penicillin V, amoxicillin, and ampicillin.
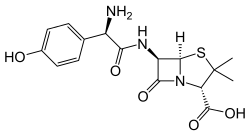
Amoxicillin is a widely used penicillin that is often combined with clavulanic acid to overcome resistance.
Cephalosporins[edit]
Cephalosporins are similar to penicillins but have a broader spectrum of activity. They are classified into generations, with each subsequent generation having increased activity against Gram-negative bacteria and better resistance to β-lactamases.
Carbapenems[edit]
Carbapenems are highly resistant to most β-lactamases and have a very broad spectrum of activity. They are often used as a last resort for treating severe infections caused by multidrug-resistant bacteria.
Monobactams[edit]
Monobactams, such as aztreonam, are effective primarily against Gram-negative bacteria and are resistant to some β-lactamases.
Resistance[edit]
Resistance to β-lactam antibiotics is a significant clinical problem. The most common mechanism of resistance is the production of β-lactamases, enzymes that hydrolyze the β-lactam ring, rendering the antibiotic ineffective.

Clavulanic acid is a β-lactamase inhibitor that is often combined with β-lactam antibiotics to overcome resistance.
Clinical Use[edit]
β-Lactam antibiotics are used to treat a wide range of infections, including pneumonia, meningitis, sepsis, and urinary tract infections. They are generally well-tolerated, but allergic reactions can occur in some individuals.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian