1,3-Dipolar cycloaddition
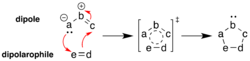
1,3-Dipolar cycloaddition is a type of cycloaddition reaction in organic chemistry where a 1,3-dipole reacts with a dipolarophile to form a five-membered ring. This reaction is a subset of the more general pericyclic reactions and is a valuable method for constructing heterocycles.
Mechanism[edit]
The mechanism of 1,3-dipolar cycloaddition involves the interaction between a 1,3-dipole and a dipolarophile. The 1,3-dipole is a molecule with a delocalized set of electrons over three atoms, typically represented as X=Y+-Z−. The dipolarophile is usually an alkene or alkyne that can participate in the cycloaddition. The reaction proceeds through a concerted mechanism, meaning that the bond formation and bond breaking occur simultaneously in a single step. This results in the formation of a five-membered ring, which can be either a pyrrolidine, isoxazoline, or other heterocyclic structures depending on the nature of the 1,3-dipole and dipolarophile.
Types of 1,3-Dipoles[edit]
Common 1,3-dipoles include:
- Azides (R-N3)
- Nitrile oxides (R-C≡N+-O−)
- Nitrile imines (R-C≡N+-N−)
- Nitrile ylides (R-C≡N+-C−)
- Ozone (O3)
Applications[edit]
1,3-Dipolar cycloaddition reactions are widely used in the synthesis of natural products, pharmaceuticals, and other complex organic molecules. They are particularly valuable for constructing heterocycles, which are common motifs in many biologically active compounds. One notable application is the synthesis of triazoles via the Huisgen cycloaddition, which involves the reaction of an azide with an alkyne. This reaction is a cornerstone of click chemistry, a concept introduced by K. Barry Sharpless.
Stereochemistry[edit]
The stereochemistry of 1,3-dipolar cycloaddition can be complex, as the reaction can produce multiple stereoisomers. The selectivity of the reaction can be influenced by the nature of the substituents on the 1,3-dipole and the dipolarophile, as well as by the reaction conditions.
See also[edit]
References[edit]
External links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian