Acid dissociation constant
Acid dissociation constant is a quantitative measure of the strength of an acid in solution. It is commonly denoted as Ka and represents the equilibrium constant for the dissociation of an acid in an acid-base reaction.
For a generic weak acid, the dissociation reaction is written as:
- HA ⇌ H+ + A−
where HA represents the undissociated acid, H+ represents the released hydrogen ion, and A− represents the conjugate base of the acid.
A larger value of Ka indicates that a greater proportion of the acid dissociates in solution. Therefore, acids with higher Ka values are generally stronger acids, while acids with lower Ka values are weaker acids.
Definition[edit]
The acid dissociation constant is defined by the equilibrium expression:
- Ka = ([H+][A−]) / [HA]
where:
- [H+] is the molar concentration of hydrogen ions
- [A−] is the molar concentration of the conjugate base
- [HA] is the molar concentration of the undissociated acid
In aqueous solution, the hydrogen ion is often represented more accurately as hydronium ion, H3O+. The reaction may therefore also be written as:
- HA + H2O ⇌ H3O+ + A−
and the expression may be written as:
- Ka = ([H3O+][A−]) / [HA]
pKa[edit]
The pKa is the negative base-10 logarithm of the acid dissociation constant:
- pKa = −log10(Ka)
A lower pKa value indicates a stronger acid, while a higher pKa value indicates a weaker acid.
For example:
- A strong acid has a very low or negative pKa
- A weak acid has a higher pKa
- Acetic acid has a pKa of approximately 4.76 in water at room temperature
Relationship to acid strength[edit]
The value of Ka reflects the position of the chemical equilibrium:
- If Ka is large, the equilibrium favors dissociation, producing more H+ and A−.
- If Ka is small, the equilibrium favors the undissociated acid, HA.
- Strong acids dissociate nearly completely in water.
- Weak acids dissociate only partially in water.
The relationship between Ka and pKa is inverse:
- Higher Ka = lower pKa = stronger acid
- Lower Ka = higher pKa = weaker acid
Factors affecting acid strength[edit]
Several chemical factors influence acid strength and therefore affect the value of Ka.
Electronegativity[edit]
An acid is generally stronger when its conjugate base is stabilized by an electronegative atom. More electronegative atoms can better stabilize negative charge.
Atomic size[edit]
For acids in which the acidic hydrogen is bonded to atoms in the same group of the periodic table, acidity often increases as atomic size increases. Larger atoms can distribute negative charge over a larger volume.
Resonance[edit]
Resonance stabilizes the conjugate base by spreading negative charge over multiple atoms. Acids whose conjugate bases are resonance-stabilized are usually stronger.
Inductive effect[edit]
Electron-withdrawing groups can stabilize the conjugate base through the inductive effect. This increases acidity. For example, chloroacetic acid is stronger than acetic acid because the chlorine atom withdraws electron density.
Hybridization[edit]
The hybridization of the atom bearing the negative charge can affect acidity. Greater s-character generally stabilizes negative charge more effectively. For example, an sp-hybridized carbon is more acidic than an sp2 or sp3 carbon.
Solvent effects[edit]
The solvent can influence acid dissociation by stabilizing ions. Water, a polar solvent, stabilizes charged species and supports acid-base dissociation.
Examples[edit]
Acetic acid[edit]
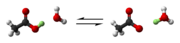
Acetic acid is a weak carboxylic acid with the formula CH3COOH. It partially dissociates in water to form acetate ions and hydrogen ions:
- CH3COOH ⇌ H+ + CH3COO−
Acetic acid has a pKa of approximately 4.76.
Phosphoric acid[edit]
Phosphoric acid is a triprotic acid, meaning it can donate three protons. It has three acid dissociation constants:
- H3PO4 ⇌ H+ + H2PO4−
- H2PO4− ⇌ H+ + HPO42−
- HPO42− ⇌ H+ + PO43−
Each step has a different Ka and pKa value.
Citric acid[edit]
Citric acid is a weak organic acid with the formula C6H8O7. It contains three carboxyl groups and is therefore a triprotic acid. Each acidic proton has a separate dissociation constant.
Carbonic acid[edit]
Carbonic acid forms when carbon dioxide dissolves in water. It plays an important role in blood pH, buffer systems, and respiration.
- H2CO3 ⇌ H+ + HCO3−
The bicarbonate buffer system is important in physiology and acid-base homeostasis.
Acid dissociation and buffers[edit]
The acid dissociation constant is important in understanding buffer solutions. A buffer resists changes in pH when small amounts of acid or base are added.
The buffering capacity is greatest when the pH of the solution is close to the pKa of the weak acid. This relationship is described by the Henderson-Hasselbalch equation:
- pH = pKa + log10([A−] / [HA])
This equation is widely used in chemistry, biochemistry, pharmacology, and medicine.
Biological and medical importance[edit]
Acid dissociation constants are important in many areas of medicine, pharmacology, and biochemistry.
They help explain:
- The ionization of drugs
- Drug absorption
- Drug distribution
- Blood pH regulation
- Enzyme function
- Protein charge
- Metabolic acidosis
- Metabolic alkalosis
- Respiratory acidosis
- Respiratory alkalosis
Many medications are weak acids or weak bases. Their degree of ionization depends on their pKa and the pH of the environment. This affects how well they cross cell membranes and how they are distributed in the body.
Common approximate pKa values[edit]
| Acid | Formula | Approximate pKa | Notes |
|---|---|---|---|
| Hydrochloric acid | HCl | −6 | Strong acid |
| Sulfuric acid | H2SO4 | −3 | Strong acid for first dissociation |
| Lactic acid | C3H6O3 | 3.86 | Important in metabolism |
| Acetic acid | CH3COOH | 4.76 | Weak carboxylic acid |
| Carbonic acid | H2CO3 | 6.35 | Important in blood buffering |
| Ammonium | NH4+ | 9.25 | Conjugate acid of ammonia |
| Water | H2O | 15.7 | Very weak acid |
Related concepts[edit]
- Acid
- Base
- Conjugate acid
- Conjugate base
- Base dissociation constant
- Chemical equilibrium
- pH
- pOH
- Buffer solution
- Titration
- Henderson-Hasselbalch equation
- Le Chatelier's principle
Gallery[edit]
-
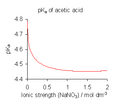
pKa of acetic acid
-
Weak acid speciation
-
Carboxylic acid dimers
-
Acetic acid pKa in dioxane-water
-
Chloroacetic acid pKa
-
Fumaric acid structure
-
Maleic acid structure
-
Proton sponge
-
Oxalic acid titration
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian