Aldol condensation
A chemical reaction involving the formation of a _-hydroxy ketone or aldehyde
Aldol condensation is an important organic reaction in which an enolate ion reacts with a carbonyl compound to form a _-hydroxy ketone or aldehyde, followed by dehydration to give a conjugated enone. This reaction is a key step in the formation of carbon-carbon bonds in organic synthesis.
Mechanism[edit]
The aldol condensation can proceed via either a base-catalyzed or an acid-catalyzed mechanism.
Base-catalyzed mechanism[edit]
In the base-catalyzed aldol condensation, a base such as hydroxide or an alkoxide ion abstracts a proton from the _-carbon of a carbonyl compound, generating an enolate ion. This enolate ion then attacks the carbonyl carbon of another molecule, forming a _-hydroxy ketone or aldehyde. Subsequent dehydration leads to the formation of an _,_-unsaturated carbonyl compound.
Acid-catalyzed mechanism[edit]
In the acid-catalyzed aldol condensation, the carbonyl compound is protonated, making it more electrophilic. An enol form of the carbonyl compound then attacks the protonated carbonyl group, leading to the formation of a _-hydroxy ketone or aldehyde. Dehydration occurs to yield the _,_-unsaturated carbonyl compound.
Examples[edit]
A classic example of an aldol condensation is the reaction between acetaldehyde molecules to form crotonaldehyde.
Applications[edit]
Aldol condensations are widely used in the synthesis of complex molecules, including natural products and pharmaceuticals. They are also employed in industrial processes, such as the production of pentaerythritol, a key component in the manufacture of alkyd resins.
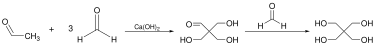
Variations[edit]
Several variations of the aldol condensation exist, including the Henry reaction, the Robinson annulation, and the Claisen-Schmidt condensation. These variations expand the scope and utility of the aldol reaction in organic synthesis.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian