Amineptine
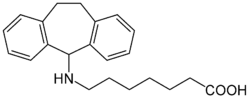
Amineptine, sold under the brand name Survector among others, is an atypical antidepressant that is no longer marketed. It was developed by the French Society of Medical Research in the 1960s.
Pharmacology[edit]
Amineptine is a tricyclic antidepressant (TCA) that selectively inhibits the reuptake of dopamine and to a lesser extent, norepinephrine, thereby increasing the levels of these neurotransmitters in the brain. Unlike other TCAs, it does not inhibit the reuptake of serotonin and does not have anticholinergic properties.
Medical uses[edit]
Amineptine was used for the treatment of major depressive disorder and dysthymia. It was noted for its activating and stimulant effects, making it useful for patients with retarded depression.
Side effects[edit]
Common side effects of amineptine include insomnia, agitation, irritability, hypomania and skin rash. Rare but serious side effects include hepatotoxicity and dependence.
History[edit]
Amineptine was developed by the French Society of Medical Research in the 1960s. It was withdrawn from the market in the late 1990s due to concerns about its potential for abuse and hepatotoxicity.
Society and culture[edit]
Amineptine was marketed under several brand names, including Survector, Maneon, Directim, and Neolior. It was available in some countries in Europe, South America, and Asia, but was never approved for use in the United States.
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
