Arylcyclohexylamine
Arylcyclohexylamine[edit]
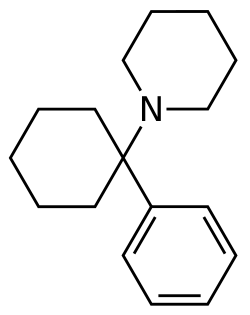
Arylcyclohexylamines are a class of dissociative anesthetics that are chemically related to phencyclidine (PCP) and ketamine. These compounds are characterized by their ability to induce a state of dissociation, where the user experiences a detachment from reality, often accompanied by hallucinations and altered perceptions of time and space.
Chemical Structure[edit]
Arylcyclohexylamines are defined by their core structure, which consists of a cyclohexane ring bound to an aryl group and an amine group. This structure is responsible for their pharmacological effects. The general formula can be represented as follows:
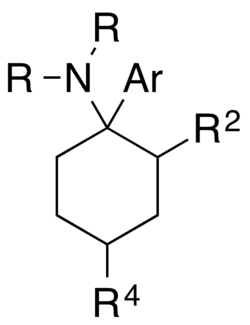
The cyclohexane ring provides the backbone of the molecule, while the aryl group and amine group contribute to the compound's activity at the NMDA receptor, which is a key target for their anesthetic and psychoactive effects.
Pharmacology[edit]
Arylcyclohexylamines primarily act as NMDA receptor antagonists. By blocking these receptors, they inhibit the action of the neurotransmitter glutamate, which is involved in excitatory signaling in the brain. This blockade leads to the dissociative effects characteristic of these compounds.
Common Arylcyclohexylamines[edit]
Several arylcyclohexylamines have been synthesized and studied for their pharmacological properties. Some of the most notable include:
- Phencyclidine (PCP): Originally developed as an anesthetic, PCP is now primarily known for its recreational use and potential for abuse.
- Ketamine: Widely used in both human and veterinary medicine as an anesthetic, ketamine is also used recreationally and has been studied for its antidepressant effects.
- Eticyclidine (PCE):
A derivative of PCP, PCE has similar effects but is less commonly encountered.
- 4-Methyl-PCP:
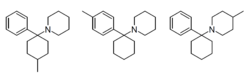
A potent analog of PCP, known for its strong dissociative effects.
- PCPr:
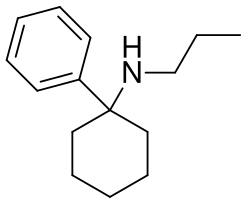
Another analog of PCP, with a longer duration of action.
- PCiP:
A less common analog with unique pharmacological properties.
Clinical and Recreational Use[edit]
Arylcyclohexylamines have been used in clinical settings primarily as anesthetics. However, their potential for abuse and the risk of adverse psychological effects have limited their use. Recreationally, these compounds are sought after for their hallucinogenic and dissociative effects, but they carry significant risks, including neurotoxicity, addiction, and psychosis.
Safety and Legal Status[edit]
The legal status of arylcyclohexylamines varies by country. Many of these compounds are controlled substances due to their potential for abuse and harmful effects. In clinical settings, their use is carefully regulated to minimize risks to patients.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian