Cephamycin
Cephamycin[edit]
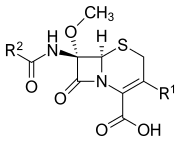
Cephamycins are a group of beta-lactam antibiotics that are structurally and functionally related to cephalosporins. They are characterized by the presence of a methoxy group at the 7-alpha position of the beta-lactam ring, which confers resistance to certain beta-lactamases produced by bacteria. This makes cephamycins particularly useful in treating infections caused by beta-lactamase-producing organisms.
Structure and Mechanism of Action[edit]
Cephamycins share a core structure with cephalosporins, which includes a beta-lactam ring fused to a dihydrothiazine ring. The distinguishing feature of cephamycins is the methoxy group at the 7-alpha position. This structural modification enhances their stability against beta-lactamase enzymes.
The mechanism of action of cephamycins, like other beta-lactam antibiotics, involves the inhibition of bacterial cell wall synthesis. They achieve this by binding to and inactivating penicillin-binding proteins (PBPs), which are essential for the cross-linking of the peptidoglycan layer of the bacterial cell wall. This leads to cell lysis and death of the bacterium.
Clinical Uses[edit]
Cephamycins are used to treat a variety of bacterial infections, particularly those caused by anaerobic bacteria and Gram-negative organisms. They are effective against organisms such as Escherichia coli, Klebsiella pneumoniae, and Bacteroides fragilis.
Commonly used cephamycins include:
These antibiotics are often used in surgical prophylaxis and in the treatment of intra-abdominal and pelvic infections.
Resistance[edit]
While cephamycins are resistant to many beta-lactamases, resistance can still occur through other mechanisms. Bacteria may acquire resistance through the production of extended-spectrum beta-lactamases (ESBLs) or through alterations in penicillin-binding proteins. Additionally, efflux pumps and changes in porin channels can contribute to resistance.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian