Ethylenediaminetetraacetic acid
Ethylenediaminetetraacetic Acid
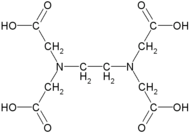
Ethylenediaminetetraacetic acid (EDTA) is a synthetic compound that is widely used in various industrial and scientific applications. It is a chelating agent, meaning that it can bind to metal ions and form stable complexes. EDTA is often used as a sequestering agent to remove metal ions from solutions or to prevent their precipitation.
Chemical Properties[edit]
EDTA is a colorless crystalline powder that is soluble in water. It has a molecular formula of C10H16N2O8 and a molar mass of 292.24 g/mol. EDTA is a polydentate ligand, meaning that it can form multiple bonds with a metal ion. It has six coordinating atoms, including four carboxylic acid groups and two nitrogen atoms from the ethylenediamine moiety.
Uses[edit]
EDTA has a wide range of applications in various industries, including food, pharmaceuticals, and cosmetics. It is used as a preservative in foods and beverages to prevent oxidation and rancidity. EDTA can also be used as a stabilizer in pharmaceuticals to improve the shelf life of drugs and prevent their degradation.
In the textile industry, EDTA is used as a color fixative and to remove metal ions that can interfere with the dyeing process. EDTA is also used in the oil industry as a scale inhibitor to prevent the buildup of calcium and magnesium ions in pipelines and equipment.
In scientific research, EDTA is used to chelate metal ions in biochemical experiments. EDTA can be used to sequester metal ions such as calcium, magnesium, and iron, which can interfere with enzyme activity or protein function.
Health and Safety[edit]
EDTA is generally considered safe for use in foods and pharmaceuticals. The US Food and Drug Administration (FDA) has approved the use of EDTA as a food additive, and it is generally recognized as safe (GRAS) when used in accordance with good manufacturing practices.
However, EDTA can be toxic in high concentrations. It can chelate essential minerals such as calcium and magnesium, leading to mineral deficiencies and other health problems. EDTA can also be irritating to the skin and eyes and can cause respiratory problems if inhaled.
Environmental Impact[edit]
EDTA is biodegradable and does not accumulate in the environment. However, it can chelate metal ions in soil and water, which can have negative effects on plant and animal life. EDTA can also leach heavy metals from soil, which can lead to contamination of groundwater.
History[edit]
EDTA was first synthesized in 1935 by Ferdinand Münz, a German chemist who was searching for a compound that could bind to and remove metal ions from blood. He discovered that EDTA could chelate calcium ions, which led to its use as an anticoagulant in blood collection tubes.
EDTA's chelating properties were further studied in the 1940s and 1950s, leading to its use in a wide range of industrial and scientific applications.
Production[edit]
EDTA is produced by reacting ethylenediamine with chloroacetic acid, which forms ethylenediaminetetraacetic acid. The reaction can be catalyzed by a base such as sodium hydroxide or sodium carbonate. The resulting product can be purified by recrystallization or ion exchange chromatography.
Analytical Chemistry[edit]
EDTA is often used in analytical chemistry to determine the concentration of metal ions in a sample. The sample is treated with EDTA, which binds to the metal ions and forms a complex. The complex can then be analyzed using techniques such as spectrophotometry or atomic absorption spectroscopy.
Chelation Therapy[edit]
EDTA has been used in chelation therapy to treat heavy metal poisoning and cardiovascular disease. Chelation therapy involves the intravenous infusion of EDTA, which binds to metal ions in the bloodstream and helps remove them from the body. Chelation therapy has been controversial, with some studies suggesting that it may be effective in treating cardiovascular disease, while others have found no significant benefit.
Conclusion[edit]
EDTA is a versatile compound that is widely used in various industrial and scientific applications. Its chelating properties make it useful for removing metal ions from solutions or as a sequestering agent. EDTA is generally considered safe when used in foods and pharmaceuticals, but can be toxic in high concentrations. EDTA's history and uses make it an important compound in the fields of chemistry and medicine.
References[edit]
See also[edit]
Chelation therapy Sequestering agent Metal ion
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian