Ester
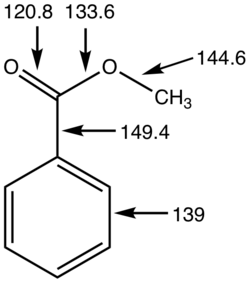
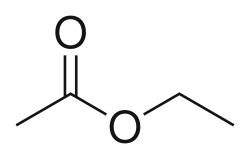
Ester is a class of chemical compounds derived from acids, in which at least one hydroxyl (-OH) group is replaced by an alkoxy (-OR) group. Esters are typically formed from the reaction between a carboxylic acid and an alcohol and are widely recognized as the most common type of carboxylic acid derivatives.
Esters are prevalent in both organic and inorganic chemistry, with broad applications ranging from flavors and fragrances to plastic production and biochemistry.
Chemical Structure[edit]
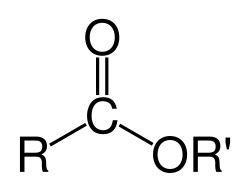
Esters contain a characteristic functional group with the general structure:
R−C(=O)−O−R′
Where:
- R = an organic group derived from the acid
- R′ = an organic group derived from the alcohol
This structure features a carbonyl group (C=O) directly bonded to an ether-like oxygen atom (O-R′). In aromatic esters, either the R or R′ group may be an aryl group (e.g., phenyl).
Formation (Esterification)[edit]
Esters are typically synthesized through a chemical reaction known as esterification. This process involves the condensation of a carboxylic acid and an alcohol, producing an ester and water as a byproduct:
R−COOH + R′−OH → R−COO−R′ + H₂O
This reaction is commonly catalyzed by strong acids, such as concentrated sulfuric acid (H₂SO₄), which acts both as a catalyst and a dehydrating agent.
Fischer Esterification[edit]
The most well-known esterification method is the Fischer esterification, discovered by Emil Fischer. It is a reversible reaction and is often driven to completion by the removal of water.
Classification[edit]
Esters can be classified based on their origin:
- Aliphatic esters – formed from aliphatic acids and alcohols (e.g., ethyl acetate)
- Aromatic esters – contain an aromatic ring in either the acid or alcohol portion (e.g., methyl benzoate)
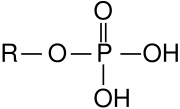
- Phosphate esters – derived from phosphoric acid, important in biochemistry (e.g., DNA backbone)
Physical and Chemical Properties[edit]
- Most esters are colorless, volatile liquids at room temperature
- They have pleasant, often fruity odors
- Esters are generally less polar than acids or alcohols
- They undergo hydrolysis in acidic or basic media (acid/base-catalyzed ester hydrolysis)
Uses[edit]
Esters are widely utilized in various industries due to their reactivity, pleasant aroma, and solubility characteristics:
- Fragrances and Flavors: Many esters have fruity smells and are used in perfumes, flavorings, and essential oils (e.g., isoamyl acetate smells like bananas)
- Solvents: Esters like ethyl acetate are used as solvents in paints, coatings, and adhesives
- Polymers and Plastics: Esters are used in producing polyesters (e.g., polyethylene terephthalate or PET)
- Biofuels: Biodiesel is composed of fatty acid methyl esters (FAMEs)
- Pharmaceuticals: Some ester derivatives function as prodrugs, improving solubility or absorption
Reactions of Esters[edit]
Esters participate in a range of chemical reactions:
- Hydrolysis: Esters can be broken down into their parent acid and alcohol under acidic or basic conditions
- Transesterification: Reaction with an alcohol to form a different ester
- Reduction: Esters can be reduced to primary alcohols using reducing agents like lithium aluminum hydride (LiAlH₄)
- Claisen condensation: Two esters react in the presence of a base to form β-keto esters
Biological Importance[edit]
In biochemistry, esters play critical roles:
- Triglycerides – formed from glycerol and fatty acids, essential for fat storage in animals
- Phosphoesters – found in DNA and RNA backbones
- Lactones – cyclic esters found in many natural products and hormones
Ester Gallery[edit]
-
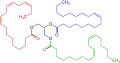
Structure of an unsaturated triglyceride (ester of glycerol)
-
Steglich esterification mechanism
-
Fischer esterification equilibrium
-
Acid and base-catalyzed ester hydrolysis
-
Claisen condensation reaction of esters
-
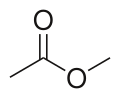
Methyl acetate (common ester)
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian