Ethamoxytriphetol
Overview of the chemical compound Ethamoxytriphetol
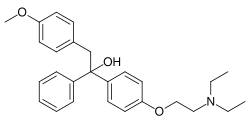
Ethamoxytriphetol, also known by its developmental code name MER-25, is a nonsteroidal selective estrogen receptor modulator (SERM) that was developed in the 1960s. It was one of the first compounds to be identified with antiestrogenic properties, and it has been used primarily in scientific research to study the effects of estrogen receptor modulation.
Chemical Structure and Properties[edit]
Ethamoxytriphetol is a triphenylethylene derivative, which is a class of compounds known for their ability to interact with estrogen receptors. The chemical structure of Ethamoxytriphetol is characterized by three phenyl rings and an ether linkage, which contribute to its binding affinity and selectivity for estrogen receptors.
Mechanism of Action[edit]
As a SERM, Ethamoxytriphetol exhibits both estrogenic and antiestrogenic effects depending on the tissue type. It acts as an antagonist in some tissues, such as the breast, where it blocks the effects of estrogen, and as an agonist in others, such as bone and the cardiovascular system, where it mimics the effects of estrogen. This dual action is due to its ability to bind to estrogen receptors and modulate their activity in a tissue-selective manner.
Pharmacological Effects[edit]
Ethamoxytriphetol was initially investigated for its potential use in treating estrogen-dependent conditions such as breast cancer and endometriosis. However, its development for clinical use was discontinued due to the availability of more effective and selective agents. Despite this, Ethamoxytriphetol remains a valuable tool in research for understanding the role of estrogen receptors in various physiological and pathological processes.
Research Applications[edit]
In scientific research, Ethamoxytriphetol has been used to study the mechanisms of estrogen receptor modulation and the effects of estrogen on different tissues. It has provided insights into the development of newer SERMs with improved efficacy and safety profiles. Researchers continue to use Ethamoxytriphetol to explore the complex interactions between estrogen receptors and their ligands.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian