Ethylene glycol dinitrate
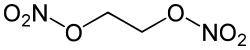
0Ethylene glycol dinitrate (EGDN, NGc, nitroglycol), is a colorless, volatile, and viscous liquid that is a powerful explosive. It is an ester of nitric acid and ethylene glycol.
History[edit]
Ethylene glycol dinitrate was first produced in 1857 by Ascanio Sobrero, but it was not until World War I that it was used on a large scale for military purposes. It was primarily used as a component in dynamite and other explosives.
Production[edit]
The production of ethylene glycol dinitrate involves the nitration of ethylene glycol with a mixture of sulfuric acid and nitric acid. This process is highly exothermic and can lead to violent reactions if not carefully controlled.
Properties[edit]
Ethylene glycol dinitrate is a colorless, oily liquid that is highly sensitive to heat, shock, and friction. It has a high explosive velocity and is more powerful than nitroglycerin. It is also highly toxic and can be absorbed through the skin, causing headaches, dizziness, and even heart and lung problems.
Uses[edit]
Ethylene glycol dinitrate is primarily used as an explosive. It is often mixed with other materials to form a more stable and safe-to-handle explosive. It is also used in the manufacture of detonators and blasting caps.
Safety[edit]
Due to its high sensitivity to shock and friction, ethylene glycol dinitrate must be handled with extreme care. It is also a strong vasodilator, and can cause headaches, dizziness, and even heart and lung problems if absorbed through the skin.
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
