Hemiacetal
Hemiacetal is a functional group in organic chemistry that consists of an alcohol and an ether connected to the same carbon atom. Hemiacetals are formed through the reaction of an aldehyde and an alcohol. They are unstable and often further react to form acetals.
Formation[edit]
Hemiacetals are formed through the reaction of an aldehyde or a ketone with an alcohol in the presence of acid. The reaction involves the addition of the alcohol to the carbonyl group, resulting in the formation of a tetrahedral intermediate. This intermediate then loses a proton to form the hemiacetal.
Properties[edit]
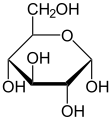
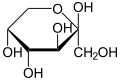
Hemiacetals are generally unstable and will further react to form acetals. However, in some cases, such as in the case of glucose, hemiacetals can be stable. This is due to the formation of a five or six-membered ring, which is energetically favorable.
Uses[edit]
Hemiacetals are important in the formation of many biological molecules. For example, glucose exists in solution primarily as a hemiacetal. They are also important in the formation of certain polymers, such as polyesters.
See also[edit]
Hemiacetal[edit]
-
Hemiacetal and Hemiketal structure
-
Formation of hemiacetals
-
Hemiketal formation
-
Lactol equilibrium
-
Robust Hemiacetals
-
Alpha-D-Glucopyranose
-
Beta-D-Fructopyranose
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian