Histone methylation

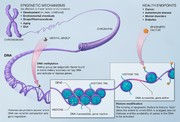
Histone methylation is a form of post-translational modification of proteins involving the addition of methyl groups to the amino acids of histone proteins. This process plays a critical role in the regulation of gene expression and is essential for the structure and function of chromatin. Histone methylation can either activate or repress gene expression, depending on the specific amino acids that are methylated and the number of methyl groups added.
Overview[edit]
Histones are proteins that package and order the DNA into structural units called nucleosomes, which make up the chromatin in eukaryotic cells. The methylation of histones can influence chromatin structure and thereby regulate access of the transcription machinery to the DNA, affecting gene expression. Histone methylation occurs on specific lysine and arginine residues on the histone tails protruding from the nucleosome. The enzymes responsible for adding methyl groups to histones are known as histone methyltransferases (HMTs), while those that remove methyl groups are called histone demethylases (HDMs).
Mechanism[edit]
Histone methylation is catalyzed by HMTs, which transfer methyl groups from S-adenosyl methionine (SAM) to the amino group of lysine or arginine residues on histones. This modification can occur in the form of mono-, di-, or tri-methylation on lysine residues and mono- or di-methylation on arginine residues. The specific pattern of methylation can influence the binding of chromatin-associated proteins, including those that promote or inhibit transcription, thereby affecting gene expression.
Function[edit]
The function of histone methylation is diverse and context-dependent. It can lead to either activation or repression of transcription, depending on the specific histone marks. For example, trimethylation of histone H3 at lysine 4 (H3K4me3) is associated with transcriptional activation, while trimethylation of histone H3 at lysine 27 (H3K27me3) is linked to transcriptional repression. These modifications serve as docking sites for protein complexes that modulate the accessibility of chromatin and, consequently, gene expression.
Regulation[edit]
The dynamic regulation of histone methylation is crucial for various cellular processes, including cell cycle progression, DNA repair, and development. The balance between the activities of HMTs and HDMs determines the methylation status of histones and thereby influences chromatin structure and function. Dysregulation of histone methylation has been implicated in several diseases, including cancer, highlighting its importance in maintaining cellular homeostasis.
Clinical Significance[edit]
Alterations in histone methylation patterns are associated with various human diseases, particularly cancer. Aberrant activity of HMTs or HDMs can lead to inappropriate silencing or activation of gene expression, contributing to tumorigenesis. As a result, these enzymes are considered potential targets for therapeutic intervention. Inhibitors of specific HMTs or HDMs are being developed and tested for their efficacy in reversing abnormal gene expression profiles in cancer and other diseases.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian