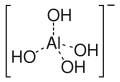Hydroxide
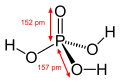
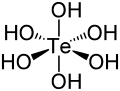
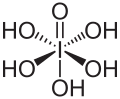
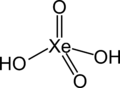
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile, and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions.
Chemical properties[edit]
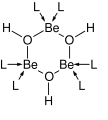
Hydroxide is a compound of an oxygen and hydrogen atom. It is a base, meaning it can accept protons (H+). Hydroxide can also act as a ligand, binding to a central atom in a molecule to form a complex. As a nucleophile, hydroxide can donate an electron pair to an electrophile. In addition, hydroxide can act as a catalyst, speeding up a chemical reaction without being consumed in the process.
Salts of hydroxide[edit]
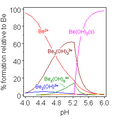
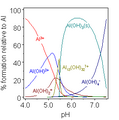
Salts of hydroxide, known as alkalis, are formed when a metal donates an electron to the hydroxide ion. Some of these salts can dissociate in water, releasing solvated hydroxide ions. This process is known as hydrolysis.
Role in water[edit]
In water, hydroxide is a minor constituent, but it plays an important role in the pH of the water. The concentration of hydroxide ions in water is equal to 10−pH, so the pH of water can be calculated from the concentration of hydroxide ions.
See also[edit]
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
-
Hydroxide
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian