Kelvin
The kelvin (symbol: K) is the base unit of temperature in the International System of Units (SI). It is named after the Scottish physicist and engineer William Thomson, 1st Baron Kelvin, who developed the concept of absolute temperature.
Definition[edit]
The kelvin is defined by taking the fixed numerical value of the Boltzmann constant, k, to be 1.380649×10⁻²³ when expressed in the unit J⋅K⁻¹, which is equal to kg⋅m²⋅s⁻²⋅K⁻¹, where the kilogram, meter, and second are defined in terms of the Planck constant, the speed of light, and the cesium frequency, respectively.
History[edit]

The concept of an absolute temperature scale was first proposed by William Thomson, 1st Baron Kelvin in 1848. He suggested that there should be a scale where absolute zero is the null point, and the degree intervals are the same as those of the Celsius scale. This led to the development of the Kelvin scale, which is now widely used in scientific contexts.
Absolute Zero[edit]
Absolute zero, 0 K, is the lowest possible temperature where nothing could be colder and no heat energy remains in a substance. At this point, the entropy of a perfect crystal is exactly zero, according to the third law of thermodynamics.
Comparison with Other Scales[edit]
The Kelvin scale is related to the Celsius scale; the only difference is the starting point. While the Celsius scale is based on the freezing and boiling points of water, the Kelvin scale starts at absolute zero. The conversion between Celsius and Kelvin is straightforward:
- K = °C + 273.15
Applications[edit]
The Kelvin scale is used in various scientific fields, including physics, chemistry, and astronomy. It is particularly useful in thermodynamics and statistical mechanics, where absolute temperature is a critical parameter.
Phase Diagrams[edit]
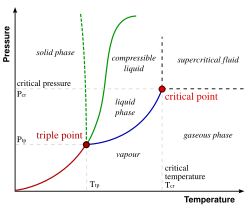
Phase diagrams often use the Kelvin scale to represent temperature. These diagrams show the conditions under which distinct phases occur and coexist at equilibrium.
SI Unit Relations[edit]
In the SI system, the kelvin is one of the seven base units. It is used to define the derived units of temperature-related quantities, such as heat capacity and thermal conductivity.
Temperature Scales Comparison[edit]
The Kelvin scale is often compared with other temperature scales, such as Fahrenheit and Celsius. While the Fahrenheit scale is commonly used in the United States, the Kelvin and Celsius scales are used worldwide in scientific contexts.
Kelvin Temperature Chart[edit]
The Kelvin temperature chart provides a visual representation of various temperatures in Kelvin, illustrating the scale's application in different scientific scenarios.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian