Michael addition reaction
The Michael addition reaction is a fundamental organic reaction that involves the addition of a nucleophile to an α,β-unsaturated carbonyl compound. This reaction is a type of nucleophilic addition and is widely used in organic synthesis to form carbon-carbon bonds. It is named after the chemist Arthur Michael, who first described the reaction in 1887.
Mechanism[edit]
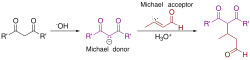
The Michael addition involves the conjugate addition of a nucleophile to an α,β-unsaturated carbonyl compound. The nucleophile, often an enolate ion, attacks the β-carbon of the α,β-unsaturated carbonyl compound, resulting in the formation of a new carbon-carbon bond. The reaction proceeds through a stepwise mechanism:
1. Formation of the Nucleophile: The nucleophile is typically generated by deprotonation of a carbon acid, such as a ketone or ester, to form an enolate ion. 2. Conjugate Addition: The enolate ion attacks the β-carbon of the α,β-unsaturated carbonyl compound, forming a new carbon-carbon bond. 3. Protonation: The resulting enolate intermediate is protonated to yield the final Michael adduct.

Applications[edit]
The Michael addition is a versatile reaction used in the synthesis of a wide range of compounds, including pharmaceuticals, natural products, and polymers. It is particularly valuable in the formation of complex molecular architectures due to its ability to form carbon-carbon bonds efficiently.
Asymmetric Michael Addition[edit]
The asymmetric Michael addition is a variant of the reaction that allows for the formation of chiral centers with high enantioselectivity. This is achieved by using chiral catalysts or chiral auxiliaries. Asymmetric Michael additions are important in the synthesis of enantiomerically pure compounds, which are crucial in the pharmaceutical industry.
Historical Context[edit]
The reaction was first reported by Arthur Michael in 1887. Michael's original work involved the addition of diethyl malonate to ethyl acrylate, demonstrating the potential of this reaction to form carbon-carbon bonds. Since then, the reaction has been extensively studied and developed, leading to numerous variations and applications.
Comparison with Other Reactions[edit]
The Michael addition is often compared to other nucleophilic addition reactions, such as the Aldol reaction and the Conrad-Limpach reaction. Each of these reactions has its own unique mechanism and applications, but the Michael addition is particularly noted for its ability to form 1,4-addition products.
Synthetic Examples[edit]
Warfarin Synthesis[edit]
The synthesis of the anticoagulant drug Warfarin involves a key Michael addition step. This reaction is used to form the core structure of the molecule, highlighting the importance of the Michael addition in pharmaceutical synthesis.
Mukaiyama-Michael Addition[edit]
The Mukaiyama-Michael addition is a variant that uses silyl enol ethers as nucleophiles. This modification allows for greater control over the reaction conditions and the formation of more complex products.
1,6-Michael Addition[edit]

The 1,6-Michael addition is a less common variant where the nucleophile adds to the δ-position of a conjugated system. This reaction expands the scope of the Michael addition to include longer conjugated systems.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian