Neodymium
Neodymium
Neodymium is a chemical element with the symbol Nd and atomic number 60. It is a soft, silvery metal that tarnishes in air. Neodymium is a member of the lanthanide series and is considered one of the rare earth elements.
Properties[edit]
Neodymium is a ductile and malleable metal. It is relatively reactive and quickly oxidizes in air, forming a pink or purple oxide layer. Neodymium has a high magnetic strength and is used in the production of powerful permanent magnets.
Applications[edit]
Magnets[edit]
Neodymium magnets, also known as NdFeB magnets, are the strongest type of permanent magnets available. They are widely used in various applications, including:
Glass and Ceramics[edit]
Neodymium is used to color glass and ceramics. It produces a range of colors from pink to purple, depending on the lighting conditions. Neodymium-doped glass is used in lasers and other optical applications.
Other Uses[edit]
Neodymium is also used in:
- Catalysts for petroleum refining
- Lighting applications
- Wind turbines
Health Effects[edit]
Neodymium is not known to be essential for any biological processes in humans. However, exposure to neodymium dust or fumes can pose health risks. Inhalation of neodymium compounds can cause lung damage and other respiratory issues.
Safety Precautions[edit]
When handling neodymium, it is important to use appropriate safety measures to prevent exposure. This includes using protective equipment and ensuring proper ventilation in work areas.
Environmental Impact[edit]
The mining and processing of neodymium can have significant environmental impacts. These include habitat destruction, water pollution, and the generation of hazardous waste. Efforts are being made to develop more sustainable methods of neodymium extraction and processing.
See Also[edit]
Chemical element with atomic number (Nd)
Neodymium, 00Nd
Neodymium[edit]
-
Neodymium(III) sulfate
-
Neodymium(III) acetate
-
Neodymium(III) hydroxide
-
Neodymium
-
Neodymium fluorescent
-
Neodymium daylight
-
Auer von Welsbach
-
Bastnaesite - Kischtimsk, Ural
-
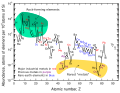
Elemental abundances
-
Monazite acid cracking process
-
Neodymag
-
Neodymium glass light bulb under fluorescent and incandescent light
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian