Nitrosation and nitrosylation
Nitrosation and nitrosylation are chemical processes involving the addition of a nitroso group (–NO) to a molecule. These reactions are significant in both organic chemistry and biochemistry, influencing the structure and function of various compounds and biological systems.
Nitrosation[edit]
Nitrosation refers to the introduction of a nitroso group into an organic compound. This process typically involves the reaction of a nitrosating agent, such as nitrous acid (HNO₂), with an amine to form a nitrosamine. Nitrosamines are a class of compounds with the general formula R₂N–NO, where R represents an organic group.
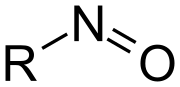
Mechanism[edit]
The mechanism of nitrosation involves the generation of a nitrosating species, often from nitrite ions (NO₂⁻) in acidic conditions. The nitrosating species then reacts with an amine to form a nitrosamine. The reaction can be represented as follows:
- R₂NH + HNO₂ → R₂N–NO + H₂O
Applications[edit]
Nitrosation is used in the synthesis of various organic compounds, including dyes, pharmaceuticals, and agrochemicals. However, nitrosamines are also known for their carcinogenic properties, making the control of nitrosation processes important in food and drug industries.
Nitrosylation[edit]
Nitrosylation involves the covalent attachment of a nitric oxide (NO) group to a metal center or a thiol group in proteins. This process is crucial in the regulation of protein function and signaling pathways in biological systems.
Mechanism[edit]
In biological systems, nitrosylation often occurs through the interaction of nitric oxide with thiol groups in cysteine residues of proteins, forming S-nitrosothiols. This modification can alter the activity, localization, and interaction of proteins.
Biological Significance[edit]
Nitrosylation plays a vital role in cellular signaling and regulation. It is involved in processes such as vasodilation, neurotransmission, and immune response. Dysregulation of nitrosylation is associated with various diseases, including cardiovascular disorders and neurodegenerative diseases.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian