Oxygen

Chemical element with symbol O and atomic number 8
Oxygen is a chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table, and is classified as a highly reactive nonmetal and a strong oxidizing agent. Oxygen readily forms oxides with most elements and many compounds.
Properties[edit]
- Atomic number: 8
- Symbol: O
- Atomic mass: 15.999 u
- Electron configuration: 1s² 2s² 2p⁴
- Standard state: Gas at room temperature
- Appearance: Colorless, odorless gas
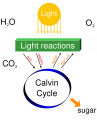
Oxygen exists in several allotropes, the most common of which is diatomic oxygen (O₂), essential for cellular respiration in most aerobic organisms. Another allotrope, ozone (O₃), plays a critical role in absorbing ultraviolet radiation in the Earth's stratosphere.
Abundance and Occurrence[edit]
Oxygen is:
- The most abundant element in the Earth's crust by mass
- The second-most abundant component of the Earth's atmosphere (approximately 21%)
- The third-most abundant element in the universe, after hydrogen and helium
It is found in water (H₂O), silicates, oxides, and in all known life forms.
Biological Role[edit]
Oxygen is essential for life in many forms:
- Required for aerobic respiration in plants, animals, fungi, and most bacteria
- Produced as a byproduct of photosynthesis in plants, algae, and cyanobacteria
Oxygen is transported in the bloodstream of vertebrates by the protein hemoglobin.
Industrial and Medical Uses[edit]
Oxygen has a wide array of applications, including:
- Medical oxygen therapy and life support systems
- Oxy-fuel welding and cutting
- Steel and chemical manufacturing
- Water treatment and environmental monitoring
- Rocket propellants and high-altitude breathing equipment
History[edit]
Oxygen was independently discovered in the 1770s by:
- Carl Wilhelm Scheele in Sweden (before 1773)
- Joseph Priestley in England (1774)
The name "oxygen" was coined by Antoine Lavoisier, who helped establish its role in combustion and respiration, disproving the phlogiston theory.
Gallery[edit]
Oxygen[edit]
-
Priestley and Fuseli
-
Oxygen molecule
-
Liquid oxygen in a beaker
-
Evolved star fusion shells
-
Sea surface oxygen
-
Phanerozoic Climate Change
-
Simple photosynthesis overview
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian