Paramethadione
An anticonvulsant medication
| Paramethadione | |
|---|---|
|
| |
| Chemical nomenclature | |
| IUPAC name | 5-ethyl-3,5-dimethyl-2,4-oxazolidinedione
|
Paramethadione is a medication used in the treatment of epilepsy, specifically for the management of absence seizures. It belongs to the class of oxazolidinediones, which are a group of anticonvulsant drugs.
Pharmacology[edit]
Paramethadione works by modulating the activity of neurons in the brain to prevent the abnormal electrical activity that leads to seizures. The exact mechanism of action is not fully understood, but it is believed to involve the stabilization of neuronal membranes and the reduction of repetitive neuronal firing.
Medical uses[edit]
Paramethadione is primarily used to treat absence seizures, which are characterized by brief, sudden lapses in attention and activity. These seizures are most common in children and are often referred to as "petit mal" seizures. Paramethadione is typically prescribed when other medications, such as ethosuximide, are not effective or cause unacceptable side effects.
Side effects[edit]
The use of paramethadione can lead to several side effects, some of which may be serious. Common side effects include drowsiness, dizziness, and gastrointestinal disturbances. More severe side effects can include blood dyscrasias, such as agranulocytosis and aplastic anemia, as well as liver toxicity. Due to these potential risks, regular monitoring of blood counts and liver function is recommended during treatment.
History[edit]
Paramethadione was developed in the mid-20th century as part of a class of anticonvulsant drugs known as oxazolidinediones. It was introduced as a treatment for absence seizures, providing an alternative to other anticonvulsants available at the time. However, due to its side effect profile, its use has declined in favor of newer medications with better safety profiles.
Chemistry[edit]
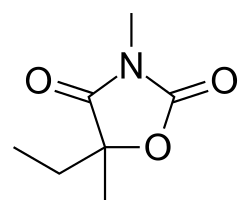
Paramethadione is chemically classified as an oxazolidinedione. Its chemical structure is characterized by a five-membered ring containing both nitrogen and oxygen atoms. The IUPAC name for paramethadione is 5-ethyl-3,5-dimethyl-2,4-oxazolidinedione, reflecting its specific molecular configuration.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian