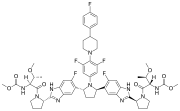
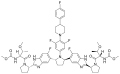
Pibrentasvir
An antiviral medication used in the treatment of hepatitis C
| Pibrentasvir | |
|---|---|
|
|
Pibrentasvir is an antiviral drug used in combination with other medications for the treatment of hepatitis C virus (HCV) infection. It is a component of the fixed-dose combination drug Glecaprevir/pibrentasvir, marketed under the brand name Mavyret.
Mechanism of Action[edit]
Pibrentasvir is a NS5A inhibitor, which means it targets the nonstructural protein 5A (NS5A) of the hepatitis C virus. NS5A is essential for viral replication and assembly, and by inhibiting this protein, pibrentasvir disrupts the life cycle of the virus, thereby reducing viral load in the patient's body.
Pharmacokinetics[edit]
Pibrentasvir is administered orally and is highly bioavailable. It is extensively metabolized in the liver and excreted primarily in the feces. The drug has a long half-life, allowing for once-daily dosing when used in combination with other antiviral agents.
Clinical Use[edit]
Pibrentasvir is used in combination with glecaprevir, a NS3/4A protease inhibitor, to treat adults with chronic hepatitis C virus infection. This combination is effective against all major genotypes of HCV, making it a pan-genotypic treatment option. The typical treatment duration is 8 to 12 weeks, depending on the patient's prior treatment history and the presence of cirrhosis.
Side Effects[edit]
Common side effects of pibrentasvir, when used in combination with glecaprevir, include headache, fatigue, and nausea. Serious side effects are rare but may include liver enzyme elevations and hypersensitivity reactions.
Contraindications[edit]
Pibrentasvir is contraindicated in patients with severe hepatic impairment (Child-Pugh C) and in those with known hypersensitivity to the drug or any of its components. Caution is advised when used in patients with moderate hepatic impairment (Child-Pugh B).
Related pages[edit]
-
Pibrentasvir
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian