Picolinic acid
Chemical compound
Picolinic acid is an organic compound with the formula C6H5NO2. It is a derivative of pyridine with a carboxylic acid substituent at the 2-position. This compound is a white solid that is soluble in water and has a slightly acidic nature.
Structure and Properties[edit]
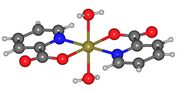
Picolinic acid is a pyridine derivative, specifically a 2-pyridinecarboxylic acid. The presence of the nitrogen atom in the pyridine ring influences its chemical properties, making it a versatile ligand in coordination chemistry. The carboxyl group can donate a proton, making picolinic acid a weak acid.
The molecular structure of picolinic acid allows it to form stable complexes with metal ions, which is significant in biological systems. The ability to chelate metals is due to the nitrogen atom in the pyridine ring and the oxygen atoms in the carboxyl group, which can coordinate with metal ions.
Synthesis[edit]
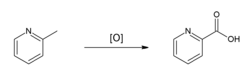
Picolinic acid can be synthesized through the oxidation of 2-picoline (2-methylpyridine). This process involves the use of oxidizing agents to convert the methyl group into a carboxylic acid group, resulting in the formation of picolinic acid. The reaction is depicted in the image showing the oxidation of 2-picoline.
Biological Role[edit]
Picolinic acid is known to play a role in the metabolism of tryptophan, an essential amino acid. It is a metabolite in the kynurenine pathway, which is the major route of tryptophan degradation in mammals. Picolinic acid has been studied for its potential role in the immune system and its ability to chelate metal ions, which may influence various biological processes.
Applications[edit]
In addition to its biological significance, picolinic acid is used in various industrial and research applications. It serves as a precursor for the synthesis of other chemical compounds and is used in the study of metal ion chelation. Its ability to form complexes with metals makes it useful in the development of metal-based drugs and in the study of metal ion transport and storage in biological systems.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian