Sodium bisulfite
Chemical compound
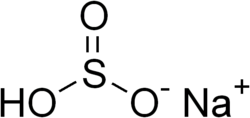
Sodium bisulfite is a chemical compound with the formula NaHSO_. It is a white solid that is soluble in water and is commonly used as a food preservative and a reducing agent in various chemical processes.
Chemical Properties[edit]
Sodium bisulfite is an inorganic compound that consists of sodium (Na_) and bisulfite (HSO__) ions. It is a sulfite salt and acts as a weak acid in aqueous solutions. The compound is known for its ability to release sulfur dioxide (SO_) when heated or in acidic conditions, which is a key feature in its use as a preservative.
Production[edit]
Sodium bisulfite is typically produced by bubbling sulfur dioxide gas through a solution of sodium carbonate (Na_CO_) or sodium hydroxide (NaOH). The reaction with sodium carbonate can be represented as:
- SO_ + Na_CO_ + H_O _ 2 NaHSO_
Uses[edit]
Sodium bisulfite is widely used in various industries due to its preservative and reducing properties.
Food Industry[edit]
In the food industry, sodium bisulfite is used as a preservative to prevent spoilage and discoloration. It is commonly added to dried fruits, wines, and other food products to maintain freshness and extend shelf life.
Chemical Industry[edit]
In the chemical industry, sodium bisulfite serves as a reducing agent in the purification of ketones and aldehydes. It is also used in the paper and pulp industry to bleach wood pulp.
Water Treatment[edit]
Sodium bisulfite is used in water treatment processes to remove excess chlorine from drinking water and wastewater. It acts as a dechlorinating agent, ensuring that water is safe for consumption and release into the environment.
Safety and Handling[edit]
Sodium bisulfite should be handled with care, as it can cause irritation to the skin, eyes, and respiratory system. It is important to use appropriate protective equipment when working with this compound.
Related Compounds[edit]
Sodium bisulfite is related to other sulfite compounds, such as sodium sulfite (Na_SO_) and sodium metabisulfite (Na_S_O_), which have similar applications and properties.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian