Sulfurous acid
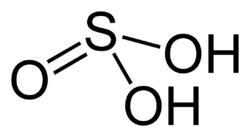
Sulfurous acid is a chemical compound with the formula H_SO_. It is an intermediate species in the formation of sulfur dioxide and sulfuric acid. Sulfurous acid is a weak and unstable acid that is not typically found in a pure form but is known to exist in aqueous solutions.
Structure and Properties[edit]
Sulfurous acid is a diprotic acid, meaning it can donate two protons (H_ ions). The molecular structure of sulfurous acid consists of a central sulfur atom bonded to two hydroxyl groups (OH) and one oxygen atom, forming a pyramidal shape. This structure is depicted in the adjacent image.
In aqueous solution, sulfurous acid partially dissociates into bisulfite (HSO__) and sulfite (SO___) ions. The equilibrium between sulfurous acid and its ions is influenced by the pH of the solution.
Formation and Reactions[edit]
Sulfurous acid is formed when sulfur dioxide (SO_) is dissolved in water:
SO_ + H_O _ H_SO_
This reaction is reversible, and sulfurous acid can decompose back into sulfur dioxide and water. In the presence of oxygen, sulfurous acid can be further oxidized to form sulfuric acid (H_SO_):
2 H_SO_ + O_ _ 2 H_SO_
Sulfurous acid acts as a reducing agent and can participate in various chemical reactions, including the reduction of iodine to iodide and the bleaching of colored substances.
Applications[edit]
Sulfurous acid and its salts, known as sulfites, are used as preservatives and antioxidants in the food and beverage industry. They help to prevent spoilage and maintain the color and flavor of foods. Sulfites are also used in the wine industry to inhibit the growth of unwanted microorganisms and to preserve the wine's freshness.
Health and Safety[edit]
Exposure to sulfurous acid and its derivatives can cause irritation to the eyes, skin, and respiratory tract. Inhalation of sulfur dioxide, which can form sulfurous acid in the respiratory system, may lead to respiratory problems, especially in individuals with asthma or other respiratory conditions.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian