Triazolopyridine
Triazolopyridine[edit]
Triazolopyridine refers to a class of heterocyclic compounds that consist of a pyridine ring fused to a triazole ring. These compounds are of significant interest in the field of medicinal chemistry due to their diverse biological activities and potential therapeutic applications.
Structure[edit]
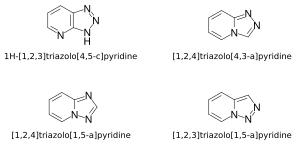
Triazolopyridines are characterized by the fusion of a six-membered pyridine ring with a five-membered triazole ring. The fusion can occur in different positions, leading to several structural isomers. The most common isomers include:
- 1,2,3-Triazolo[4,5-b]pyridine
- 1,2,3-Triazolo[1,5-a]pyridine
- 1,2,4-Triazolo[4,3-a]pyridine
Each isomer exhibits unique chemical properties and biological activities, making them valuable scaffolds in drug design.
Synthesis[edit]
The synthesis of triazolopyridines can be achieved through various methods, including:
- Cyclization reactions involving pyridine derivatives and azide compounds.
- Transition metal-catalyzed reactions, such as copper-catalyzed azide-alkyne cycloaddition (CuAAC).
- Direct functionalization of pyridine rings followed by cyclization.
These synthetic routes allow for the introduction of various substituents, enabling the fine-tuning of the compound's properties.
Biological Activity[edit]
Triazolopyridines have been studied for their potential as therapeutic agents due to their ability to interact with various biological targets. Some of the notable activities include:
- Antimicrobial activity
- Anticancer properties
- Anti-inflammatory effects
- Antiviral activity
The versatility of the triazolopyridine scaffold allows for the development of compounds with specific activity profiles, making them promising candidates in drug discovery.
Applications[edit]
In addition to their medicinal applications, triazolopyridines are used in:
- Material science for the development of novel materials with unique electronic properties.
- Agricultural chemistry as potential agrochemicals.
- Catalysis as ligands in various catalytic processes.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian