Wilkinson's catalyst
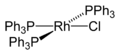
Wilkinson's catalyst, chemically known as chloridotris(triphenylphosphine)rhodium(I), is a coordination compound with the formula RhCl(PPh3)3. It is a homogeneous catalyst used in the hydrogenation of alkenes. The catalyst was first synthesized by the chemist Geoffrey Wilkinson, a Nobel Laureate, and its development marked a significant advancement in organometallic chemistry and homogeneous catalysis.
Structure and Properties[edit]
Wilkinson's catalyst is a square planar complex consisting of a rhodium center coordinated to three triphenylphosphine (PPh3) ligands and one chloride ion. This configuration provides the catalyst with its characteristic reactivity and selectivity in chemical reactions. The compound is soluble in organic solvents such as benzene, chloroform, and ethanol, which facilitates its use in homogeneous catalysis.
Synthesis[edit]
The synthesis of Wilkinson's catalyst involves the reaction of rhodium(III) chloride with an excess of triphenylphosphine in a boiling alcohol solution. The reaction proceeds via the reduction of rhodium(III) to rhodium(I) and the formation of the complex:
RhCl3·3H2O + 3 PPh3 → RhCl(PPh3)3 + 3 H2O + 2 HCl
Mechanism of Action[edit]
In the hydrogenation of alkenes, Wilkinson's catalyst facilitates the addition of hydrogen (H2) across the carbon-carbon double bond. The mechanism involves oxidative addition of H2 to the rhodium(I) center to form a rhodium(III) dihydride species. This is followed by coordination of the alkene to the metal center, insertion of the alkene into one of the Rh-H bonds (hydrometalation), and reductive elimination of the alkane from the metal. The cycle is completed when the catalyst is regenerated.
Applications[edit]
Wilkinson's catalyst is primarily used in the hydrogenation of alkenes to alkanes. It exhibits high selectivity, allowing for the hydrogenation of terminal alkenes in the presence of internal alkenes. Additionally, it has been employed in the hydrogenation of other unsaturated compounds, including aldehydes, ketones, and nitriles. Its ability to selectively hydrogenate less reactive bonds under mild conditions makes it a valuable tool in organic synthesis.
Safety and Environmental Considerations[edit]
As with many organometallic compounds, Wilkinson's catalyst should be handled with care. It is sensitive to air and moisture, requiring storage under an inert atmosphere. Proper safety equipment, such as gloves and eye protection, should be used when handling the compound. Disposal should follow regulations for hazardous waste, considering its potential environmental impact.
See Also[edit]
References[edit]
-
Wilkinson's catalyst
-
Wilkinson's catalyst
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian