(S,S)-Tetrahydrochrysene
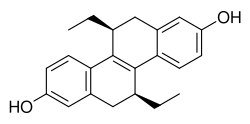
(S,S)-Tetrahydrochrysene is a synthetic compound that belongs to the class of organic molecules known as tetrahydrochrysenes. It is an enantiomer of tetrahydrochrysene, which means it is one of two stereoisomers that are mirror images of each other but cannot be superimposed. Specifically, (S,S)-tetrahydrochrysene refers to the stereoisomer with both chiral centers in the S configuration. This compound is of interest in the field of chemistry and pharmacology due to its potential applications in the study of estrogen receptors.
Chemistry[edit]
(S,S)-Tetrahydrochrysene is characterized by its unique chemical structure, which includes a polycyclic hydrocarbon backbone. The designation (S,S) indicates the specific spatial arrangement of atoms around its chiral centers, which significantly influences the compound's interactions with biological molecules. The synthesis of (S,S)-tetrahydrochrysene involves complex organic synthesis techniques, aiming to achieve the desired stereochemistry with high purity.
Pharmacology[edit]
In pharmacological studies, (S,S)-tetrahydrochrysene has been explored for its ability to bind to estrogen receptors, which are a group of proteins found inside cells that are activated by the hormone estrogen. Estrogen receptors are involved in numerous physiological processes, including reproductive system development, bone density regulation, and the cardiovascular system's function. Compounds that can modulate the activity of estrogen receptors are of significant interest for developing therapeutic agents for conditions such as osteoporosis, breast cancer, and menopausal symptoms.
Potential Applications[edit]
The interest in (S,S)-tetrahydrochrysene and its analogs lies in their potential to act as selective estrogen receptor modulators (SERMs). SERMs are a class of drugs that can either activate or block estrogen receptors in different tissues, offering the possibility to selectively mimic or inhibit estrogen's effects in the body. This selectivity allows for the development of drugs that can, for example, prevent bone loss without stimulating the growth of estrogen-sensitive tumors.
Research and Development[edit]
Research on (S,S)-tetrahydrochrysene is ongoing, with studies focusing on its binding affinity, selectivity, and efficacy in modulating estrogen receptor activity. The development of new synthetic methods has also been a key area of interest, aiming to improve the yield and purity of (S,S)-tetrahydrochrysene for research and potential therapeutic applications.
Conclusion[edit]
(S,S)-Tetrahydrochrysene represents a fascinating area of research in medicinal chemistry, with its potential applications in the development of new therapies targeting estrogen receptor-related conditions. As research progresses, it may lead to the discovery of novel drugs that can offer improved outcomes for patients with fewer side effects.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
