16-Ketoestradiol
16-Ketoestradiol[edit]
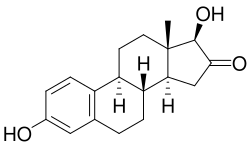
16-Ketoestradiol is a metabolite of the estrogen hormone estradiol. It is one of the many hydroxylated derivatives of estradiol that are formed during the metabolism of estrogens in the human body. This compound is of interest in the study of estrogen metabolism and its implications in various physiological and pathological processes.
Chemical Structure[edit]
16-Ketoestradiol is characterized by the presence of a keto group at the 16th position of the estradiol molecule. This modification alters the chemical properties and biological activity of the compound compared to its parent hormone, estradiol.
Biological Role[edit]
As a metabolite of estradiol, 16-Ketoestradiol is involved in the complex pathways of estrogen metabolism. Estrogens are primarily metabolized in the liver, where they undergo various enzymatic transformations, including hydroxylation and oxidation. These processes result in the formation of multiple metabolites, each with distinct biological activities.
Metabolic Pathways[edit]
The formation of 16-Ketoestradiol involves the action of specific enzymes that catalyze the oxidation of estradiol. This process is part of the broader estrogen metabolism pathway, which includes the conversion of estradiol to other metabolites such as estrone and estriol.
Clinical Significance[edit]
Understanding the metabolism of estrogens, including the formation of 16-Ketoestradiol, is important in the context of hormone replacement therapy, breast cancer, and other estrogen-related conditions. The balance and levels of different estrogen metabolites can influence the risk and progression of these conditions.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian