2-Hydroxyestrone
2-Hydroxyestrone[edit]
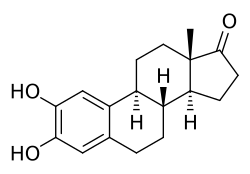
2-Hydroxyestrone is a metabolite of the estrogen hormone estrone. It is one of the major catechol estrogens, which are hydroxylated derivatives of estrogens. The hydroxylation occurs at the C2 position of the steroid ring structure, resulting in the formation of 2-hydroxyestrone.
Biochemistry[edit]
2-Hydroxyestrone is formed from estrone through the action of the enzyme cytochrome P450 1A1 (CYP1A1) and other related enzymes. This hydroxylation process is part of the phase I metabolism of estrogens, which prepares them for further metabolism and excretion.
Once formed, 2-hydroxyestrone can undergo further metabolism to form 2-methoxyestrone through the action of the enzyme catechol-O-methyltransferase (COMT). This methylation step is important for the detoxification and elimination of catechol estrogens from the body.
Biological Activity[edit]
2-Hydroxyestrone has been studied for its potential role in modulating estrogenic activity in the body. Unlike estrone and estradiol, which are potent estrogens, 2-hydroxyestrone has relatively weak estrogenic activity. It is thought to act as an antiestrogen in some tissues, potentially influencing the risk of estrogen-related diseases such as breast cancer.
Clinical Significance[edit]
The balance between different estrogen metabolites, including 2-hydroxyestrone and 16_-hydroxyestrone, has been investigated for its potential implications in health and disease. A higher ratio of 2-hydroxyestrone to 16_-hydroxyestrone is sometimes considered favorable, as 2-hydroxyestrone is thought to be less carcinogenic.
Related Compounds[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian