4-Methoxyestriol
4-Methoxyestriol[edit]
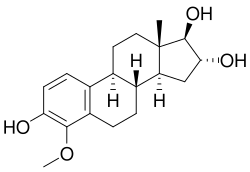
4-Methoxyestriol is a methoxylated derivative of the endogenous estrogen estriol. It is a member of the estrogen family of steroid hormones and is characterized by the presence of a methoxy group at the 4-position of the estriol molecule.
Chemical Structure[edit]
4-Methoxyestriol is a steroid with the chemical formula C19H26O4. The structure consists of a steroid nucleus with three hydroxyl groups and one methoxy group. The methoxy group is attached to the fourth carbon of the aromatic A-ring, distinguishing it from other estriol derivatives.
Biosynthesis and Metabolism[edit]
4-Methoxyestriol is synthesized from estriol through the action of specific enzymes that add a methoxy group to the 4-position. This modification can alter the biological activity and metabolism of the compound compared to estriol.
Biological Activity[edit]
As an estrogen, 4-Methoxyestriol can bind to estrogen receptors and exert estrogenic effects in the body. However, the presence of the methoxy group may influence its affinity for these receptors and its overall potency compared to other estrogens like estradiol and estriol.
Clinical Significance[edit]
The clinical significance of 4-Methoxyestriol is not as well-studied as other estrogens. However, its unique structure may offer insights into the development of selective estrogen receptor modulators (SERMs) or other therapeutic agents that target estrogen pathways.
Related Compounds[edit]
4-Methoxyestriol is related to other methoxylated estrogens and estriol derivatives. These compounds are of interest in endocrinology and pharmacology for their potential roles in hormone replacement therapy and cancer treatment.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian