Alkane
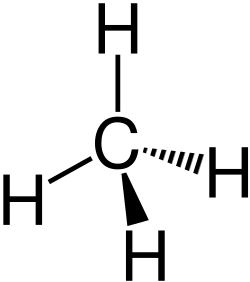
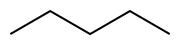
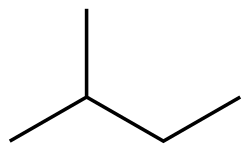
Alkanes are a class of hydrocarbons that consist entirely of single bonds and are saturated with hydrogen atoms. They are the simplest type of organic compounds and form a homologous series with a general molecular formula of CnH2n+2, where n is the number of carbon atoms. Alkanes are known for their chemical inertness and serve as a major component of natural gas and petroleum.
Structure and Nomenclature[edit]
Alkanes have a straightforward structure where carbon atoms are linked by single bonds in a chain. This chain can be straight (linear alkanes) or branched. The simplest alkane is methane (CH4), followed by ethane (C2H6), propane (C3H8), and so on. The nomenclature of alkanes follows the IUPAC (International Union of Pure and Applied Chemistry) system, which names compounds based on the length of the carbon chain and the position of any branches.
Physical Properties[edit]
The physical properties of alkanes vary with the number of carbon atoms. Lower alkanes are gases at room temperature, mid-range alkanes are liquids, and higher alkanes are waxy solids. The boiling and melting points of alkanes increase with molecular weight due to the increased van der Waals forces as the surface area of the molecules increases. However, branching in the carbon chain tends to lower these points because it reduces the surface area and thus the van der Waals forces.
Chemical Properties[edit]
Alkanes are relatively unreactive due to the strength of the carbon-carbon and carbon-hydrogen bonds. They do not easily undergo addition reactions because they lack a functional group. However, they can participate in substitution reactions, such as halogenation, under certain conditions. Alkanes can also undergo combustion, reacting with oxygen to produce carbon dioxide, water, and heat.
Uses[edit]
Alkanes are primarily used as fuels. Methane is a major component of natural gas, while other alkanes are found in petroleum and used as gasoline, diesel, and jet fuel. Alkanes are also used as solvents and as starting materials for the synthesis of more complex organic compounds.
Environmental Impact[edit]
The combustion of alkanes contributes to air pollution and the greenhouse effect. Methane, in particular, is a potent greenhouse gas. The extraction and refining of alkanes from natural sources also have environmental impacts, including habitat destruction and oil spills.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
