Ethane
Ethane is a chemical compound with the formula C2H6. It is a colorless, odorless, gaseous hydrocarbon that falls under the category of alkanes. Ethane is primarily used as a feedstock for ethylene production in the petrochemical industry.
Chemical Properties[edit]
Ethane is the second member of the alkane series and consists of two carbon atoms connected by a single bond, with each carbon atom also bonded to three hydrogen atoms. The chemical formula of ethane is C2H6. It is a nonpolar molecule due to the symmetrical arrangement of the hydrogen atoms around the two carbon atoms.
Physical Properties[edit]
Ethane is a colorless and odorless gas at standard temperature and pressure. It has a boiling point of -88.6 degrees Celsius and a melting point of -183.3 degrees Celsius. Ethane is slightly soluble in water and more soluble in organic solvents.
Production[edit]
Ethane is produced on a large scale from natural gas by fractional distillation. It can also be produced by the cracking of larger hydrocarbons in petroleum refining.
Uses[edit]
The primary use of ethane is in the production of ethylene for making plastics. It is also used as a fuel in some power plants and as a refrigerant.
Health Effects[edit]
Exposure to high levels of ethane can cause dizziness, nausea, and loss of consciousness. Long-term exposure can lead to weight loss, kidney damage, and anemia.
See Also[edit]
Ethane[edit]
-
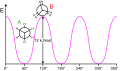
Ethane conformations and relative energies
-
Titan North Pole Lakes
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian