Barr body

Barr body
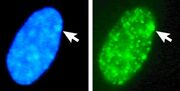
A Barr body (also known as a sex chromatin) is an inactivated X chromosome in a female somatic cell. It is named after its discoverer, Murray Barr, a Canadian physician and researcher. The presence of a Barr body is a key indicator of the genetic sex of an individual, as it is typically found in females but not in males.
Discovery and History[edit]
The Barr body was first identified by Murray Barr and his graduate student Ewart Bertram in 1949. They observed a dark-staining spot in the nuclei of female cat cells, which was later determined to be an inactivated X chromosome.
Formation[edit]
In females, who have two X chromosomes (XX), one of the X chromosomes becomes inactivated during early embryonic development through a process called X-inactivation or lyonization. This process ensures that females, like males (who have one X and one Y chromosome), have one functional copy of the X chromosome in each somatic cell. The inactivated X chromosome condenses into a compact structure known as a Barr body.
Function[edit]
The primary function of the Barr body is to regulate the dosage of X-linked genes. Without X-inactivation, females would produce twice the amount of X-linked gene products as males, which could be detrimental. The inactivation of one X chromosome equalizes the expression of X-linked genes between males and females.
Clinical Significance[edit]
The presence or absence of a Barr body can be used in cytogenetics to diagnose certain genetic conditions. For example, individuals with Turner syndrome (45,X) typically lack a Barr body, while those with Klinefelter syndrome (47,XXY) have one Barr body. The study of Barr bodies is also important in understanding disorders of sexual development and other chromosomal abnormalities.
Related Concepts[edit]
- X-inactivation
- Lyon hypothesis
- Sex chromosome
- Turner syndrome
- Klinefelter syndrome
- Dosage compensation
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian