Chanoclavine
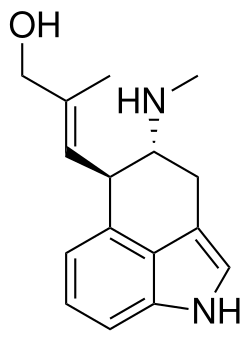
Chanoclavine is a naturally occurring ergoline alkaloid found in certain species of fungi, particularly those belonging to the genus Claviceps. It is one of the many compounds produced by these fungi, which are known for their role in the production of ergot alkaloids.
Structure and Properties[edit]
Chanoclavine has a complex molecular structure characteristic of the ergoline family. The structure includes a tetracyclic ring system with nitrogen atoms, which is a hallmark of ergoline alkaloids. The molecular formula of chanoclavine is C16H18N2O.
Biosynthesis[edit]
Chanoclavine is synthesized by fungi through a series of enzymatic reactions starting from the amino acid tryptophan. The biosynthetic pathway involves several steps, including the formation of dimethylallyltryptophan, which is then converted into chanoclavine through a series of intermediate compounds.
Biological Activity[edit]
Chanoclavine exhibits various biological activities, including interactions with serotonin receptors. However, its exact role and effects in biological systems are still under investigation. It is one of the precursors in the biosynthesis of more complex ergot alkaloids, which have significant pharmacological properties.
Applications[edit]
While chanoclavine itself is not widely used in medicine, its derivatives and related compounds have important applications. Ergot alkaloids, for instance, are used in the treatment of migraines and to induce labor in childbirth due to their ability to constrict blood vessels and stimulate uterine contractions.
Related Compounds[edit]
Chanoclavine is related to several other ergoline alkaloids, including:
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian