Clonal selection
Concept in immunology
Overview[edit]
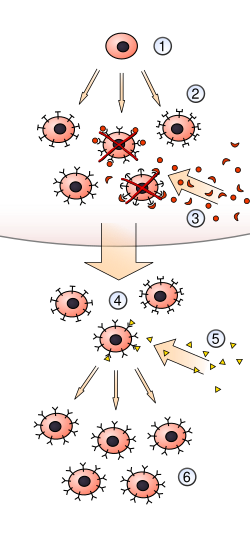
The clonal selection theory is a fundamental concept in immunology that explains how the immune system responds to antigens. It was first proposed by Frank Macfarlane Burnet in 1957. The theory describes how lymphocytes, a type of white blood cell, are selected for proliferation based on their ability to bind to specific antigens.
Mechanism[edit]
The clonal selection process begins when an antigen enters the body and is recognized by a specific B cell or T cell receptor. Each lymphocyte carries a unique receptor that is specific to a particular antigen. When an antigen binds to a receptor, it triggers the activation of the lymphocyte.
Activation and Proliferation[edit]
Upon activation, the lymphocyte undergoes rapid cell division, producing a clone of identical cells. This process is known as clonal expansion. The expanded clone consists of effector cells that actively participate in the immune response and memory cells that provide long-term immunity.
Differentiation[edit]
The effector cells differentiate into plasma cells in the case of B cells, which produce antibodies specific to the antigen. T cells differentiate into various types of effector T cells, such as cytotoxic T cells and helper T cells, which perform different functions in the immune response.
Importance[edit]
Clonal selection is crucial for the adaptive immune response, allowing the immune system to specifically target and eliminate pathogens. It also forms the basis for immunological memory, enabling a faster and more effective response upon subsequent exposure to the same antigen.
Applications[edit]
The principles of clonal selection are applied in various medical and scientific fields, including the development of vaccines and monoclonal antibodies. Understanding this process is essential for advancing immunotherapy and autoimmune disease treatments.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian