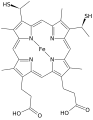
Cytochrome c
Cytochrome c is a small heme protein found loosely associated with the inner membrane of the mitochondrion. It belongs to the cytochrome c family of proteins and plays a major role in cell apoptosis and the electron transport chain.
Structure[edit]
Cytochrome c has a primary structure consisting of a chain of about 100 amino acids. Many amino acids in cytochrome c are post-translationally modified, in a process called isomerization. The protein has a secondary structure which folds into a compact tertiary structure, due to the heme group which is attached to the protein.
Function[edit]
Cytochrome c plays a key role in the electron transport chain, in the third stage of cellular respiration. It accepts electrons from the bc1 complex and transfers them to the cytochrome c oxidase complex. In addition, cytochrome c is also involved in initiation of apoptosis. Upon release of cytochrome c to the cytoplasm, the protein binds Apaf-1 and ATP, which then bind procaspase-9 to create a protein complex known as an apoptosome.
Clinical significance[edit]
Alterations in the structure or function of cytochrome c can lead to a variety of diseases, including cancer, neurodegenerative diseases, and cardiovascular diseases. For example, mutations in cytochrome c can disrupt the function of the electron transport chain, leading to a buildup of reactive oxygen species and cell damage.
See also[edit]
References[edit]
Cytochrome_c[edit]
-
Cytochrome c image 2
-
Tunafish cytochrome c crystals grown in microgravity
-
Heme c
-
Removal of O2- and H2O2 by cytochrome c
-
Peroxynitrous acid 2D
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian