Calcipotriol
What is Calcipotriol?[edit]
- Calcipotriol is a vitamin D derivative drug that slows the production of excessive skin cells.
'
What are the uses of this medicine?[edit]
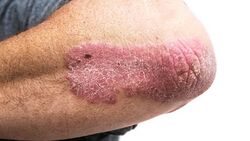
- Calcipotriol is used to treat psoriasis which is a skin disease that causes the formation of red, scaly patches due to increase production of skin cells.
How does this medicine work?[edit]
- Calcipotriol binds to the vitamin D receptor found in T-cells, which play a role in psoriasis.
- The binding controls the amount of T-cells that are differentiated and proliferated.
Who Should Not Use this medicine?[edit]
- Pregnant women
- Children
- Inform your doctors if you have any other health conditions before taking this medication.
Is this medicine FDA approved?[edit]
- This medication is FDA approved.
How should this medicine be used?[edit]
- Calcipotriol comes in 4 forms: ointment, foam, cream and liquid solution.
- It is applied twice a dice around the same time.
- The directions for cream, ointment, foam, and solution differ, so carefully read the back of the medication prior to usage and follow the instructions carefully.
- Follow your doctor's prescription and do not use more or less.
- Always wash your hands before applying the medication.
What are the dosage forms and brand names of this medicine?[edit]
- Dosage may vary depending on form of medication, brand, and your condition.
- Follow the dosage information provided on the medication or ask your doctor for personalized instructions.
Brand names:
- Dovonex®
- Sorilux®
- Calcitrene
- Daivobet®
- Enstilar®
What side effects can this medication cause?[edit]
Common side effects of Calcipotriol:
Serious side effects. Call your doctor if you experience:
- rash
- irritation of the treated or nearby area of skin
- Call your doctor if any of the side effects mentioned persists.
- This drug may also cause other side effects not listed above.
- Reach out to your doctor if you experience any other side effects while taking this medication.
What special precautions should I follow?[edit]
- Before taking this medication, inform your doctor of all the medication you are currently on and all other medical conditions you have.
- Calcipotriol causes light sensitivity, so avoid prolonged exposure to sun. Wear sunscreen, hats, and sunglasses as a precaution before going outside.
- Calcipotriol is flammable so do not use near heat or open flame.
- Do not use on face.
Do not use cosmetics or skincare products on the treated area without informing your doctor.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- The safety and efficacy of Calcipotriol during pregnancy has not been established. Inform your doctor if you are pregnant or breastfeeding.
Can this medicine be used in children?[edit]
- The safety and efficacy of Calcipotriol in children has not been established.
What should I know about storage and disposal of this medication?[edit]
- Store this medication in the container that come in with the lid tightly closed at room temperature and away from excess heat and moisture (avoid storing in bathroom).
- Keep this medication out of reach of children and pets.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian