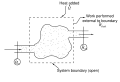
Enthalpy
Enthalpy is a concept used in thermodynamics to understand the total heat content of a system. It is a state function that depends only on the prevailing equilibrium state identified by the system's internal energy, pressure, and volume. It is an extensive quantity.
Definition[edit]
The enthalpy of a system is defined as the sum of its internal energy U and the product of its pressure P and volume V. The equation for enthalpy H is:
- H = U + PV
Understanding Enthalpy[edit]
Enthalpy is a measure of the total energy of a thermodynamic system. It includes the internal energy, which is the energy required to create a system, and the amount of energy required to make room for it by displacing its environment and establishing its volume and pressure.
Enthalpy Change[edit]
The change in enthalpy (ΔH) of a reaction is a useful quantity, as it is directly measurable. It is the energy transferred from the system to its surroundings, or vice versa, in a process that takes place at constant pressure.
Enthalpy in Chemical Reactions[edit]
In chemical reactions, the enthalpy of reaction indicates whether a reaction is exothermic (releases heat, ΔH < 0) or endothermic (absorbs heat, ΔH > 0).
See Also[edit]
-
First law of thermodynamics for an open system
-
T-s diagram of nitrogen
-
Schematic of a throttling process
-
Schematic of a compressor
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian