Eteplirsen
What is Eteplirsen?[edit]
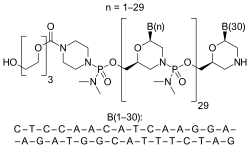
- Eteplirsen (EXONDYS 51) is an antisense oligonucleotide used for the treatment of Duchenne muscular dystrophy (DMD).
What are the uses of this medicine?[edit]
- This medicine is used for the treatment of Duchenne muscular dystrophy (DMD) in patients who have a confirmed mutation of the DMD gene that is amenable to exon 51 skipping.
- This indication is approved under accelerated approval based on an increase in dystrophin in skeletal muscle observed in some patients treated with EXONDYS 51.
How does this medicine work?[edit]
- Eteplirsen is designed to bind to exon 51 of dystrophin pre-mRNA, resulting in exclusion of this exon during mRNA processing in patients with genetic mutations that are amenable to exon 51 skipping.
- Exon skipping is intended to allow for production of an internally truncated dystrophin protein, which was evaluated in Study 2 and Study 3.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
- No clinically important drug interactions have been observed with EXONDYS 51.
Is this medicine FDA approved?[edit]
- It was approved for use in the United States in 2016.
How should this medicine be used?[edit]
Recommended Dosage:
- The recommended dose of EXONDYS 51 is 30 milligrams per kilogram administered once weekly as a 35 to 60 minute intravenous infusion via an in-line 0.2 micron filter.
Administration
- Dilution required prior to administration.
- Application of a topical anesthetic cream to the infusion site prior to administration of EXONDYS 51 may be considered.
- EXONDYS 51 is administered via intravenous infusion. Flush the intravenous access line with 0.9% Sodium Chloride Injection, USP, prior to and after infusion.
- Infuse the diluted EXONDYS 51 solution over 35 to 60 minutes via an in-line 0.2 micron filter. Do not mix other medications with EXONDYS 51 or infuse other medications concomitantly via the same intravenous access line.
- If a hypersensitivity reaction occurs, consider slowing the infusion or interrupting the EXONDYS 51 therapy
- If a dose of EXONDYS 51 is missed, it may be administered as soon as possible after the scheduled time.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form: As Injection:
- 100 mg/2 mL (50 mg/mL) in single-dose vial
- 500 mg/10 mL (50 mg/mL) in single-dose vial
This medicine is available in fallowing brand namesː
- EXONDYS 51
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- Balance disorder
- Vomiting
- Contact dermatitis
What special precautions should I follow?[edit]
- Hypersensitivity reactions, including rash and urticaria, pyrexia, flushing, cough, dyspnea, bronchospasm, and hypotension, have occurred in patients who were treated with EXONDYS 51. If a hypersensitivity reaction occurs, institute appropriate medical treatment and consider slowing the infusion or interrupting the EXONDYS 51 therapy.
What to do in case of emergency/overdose?[edit]
- There is no experience with overdose of EXONDYS 51.
Can this medicine be used in pregnancy?[edit]
- There are no human or animal data available to assess the use of EXONDYS 51 during pregnancy.
Can this medicine be used in children?[edit]
- EXONDYS 51 is indicated for the treatment of Duchenne muscular dystrophy (DMD) in patients who have a confirmed mutation of the DMD gene that is amenable to exon 51 skipping, including pediatric patients.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredient:
- eteplirsen
Inactive ingredients:
- sodium chloride
- potassium chloride
- potassium phosphate, monobasic
- sodium phosphate, dibasic, anhydrous
- sodium hydroxide
- hydrochloric acid
- water
Who manufactures and distributes this medicine?[edit]
- Packager: Sarepta Therapeutics, Inc.
What should I know about storage and disposal of this medication?[edit]
- Store EXONDYS 51 at 2°C to 8°C (36°F to 46°F).
- Do not freeze.
- Protect from light and store EXONDYS 51 in the original carton until ready for use.
| Other drugs for disorders of the musculo-skeletal system (M09) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian