Europium(III) oxide
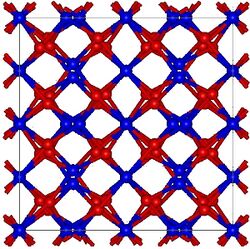
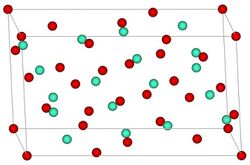
Europium(III) oxide, also known as europia, is a chemical compound composed of europium and oxygen with the formula Eu2O3. It is one of the most reactive rare earth oxides, and it serves as an important material in various applications due to its unique properties, including phosphorescence and magnetism. This compound is a part of the lanthanide series in the periodic table, which is a group of 15 chemically similar elements ranging from lanthanum to lutetium, plus scandium and yttrium.
Properties[edit]
Europium(III) oxide has distinctive physical and chemical properties. It is a white to slightly pinkish powder, insoluble in water but soluble in acids. This oxide is known for its luminescent properties, emitting bright red light when exposed to certain wavelengths, which makes it valuable in the manufacturing of phosphors for television screens, fluorescent lamps, and LEDs. Additionally, it exhibits magnetic properties at low temperatures, making it of interest in the field of cryogenics and in the development of various magnetic materials.
Synthesis[edit]
Europium(III) oxide can be synthesized through several methods. One common approach involves the calcination of europium(III) nitrate, Eu(NO3)3, or europium(III) carbonate, Eu2(CO3)3, at high temperatures. Another method is the thermal decomposition of europium oxalate, Eu2(C2O4)3. These processes result in the formation of pure europium(III) oxide powder.
Applications[edit]
The applications of europium(III) oxide are diverse and significant in various high-tech fields. In the electronics industry, it is used as a red phosphor in television screens and fluorescent lamps. Its ability to absorb neutrons makes it useful as a shielding material in nuclear reactors. Furthermore, europium-doped materials are employed in lasers and other optical devices due to their ability to emit light at specific wavelengths.
Safety[edit]
Handling europium(III) oxide requires caution. Although it is not highly toxic, it is a dust irritant, and prolonged exposure can cause skin and eye irritation. Therefore, appropriate safety measures, such as wearing gloves and eye protection, should be taken when working with this compound.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
