Fischer projection
The Fischer projection is a method or a chemical notation used to represent molecules in organic chemistry by projecting their three-dimensional (3D) structures onto a two-dimensional (2D) plane. Named after the German chemist Emil Fischer, who developed this notation in 1891, the Fischer projection is particularly useful for depicting carbohydrates and amino acids, which are molecules that have multiple chiral centers.
In a Fischer projection, the molecule is oriented so that the carbon chain runs vertically, with the most oxidized end (typically the aldehyde or carboxylic acid group in sugars) at the top. Horizontal lines represent bonds that project forward (out of the plane of the paper), while vertical lines represent bonds that project backward (into the plane of the paper). This allows for a clear representation of the stereochemistry at each chiral center, which is crucial for understanding the molecule's properties and reactivity.
The main advantage of the Fischer projection is its simplicity and ease of use, especially when comparing the stereochemistry of molecules. It is particularly helpful in the study of biochemistry and organic synthesis, where the configuration of chiral centers plays a significant role in the function and synthesis of molecules.
However, it is important to note that while Fischer projections are useful for visualizing molecules in two dimensions, they do not accurately represent the true 3D structure of molecules. Other representations, such as the Haworth projection for sugars and the chair conformation for six-membered rings, provide a more accurate depiction of the spatial arrangement of atoms.
-
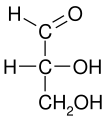
D-Glyceraldehyde Fischer Projection
-
Fischer Projection Example
-
Fischer Projection of a Molecule
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian