Fluroxene
Fluroxene[edit]
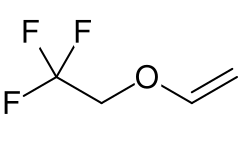
Fluroxene is a volatile inhalational anesthetic that was used in the past for the induction and maintenance of general anesthesia. It is a halogenated ether, specifically a fluorinated ethyl vinyl ether, which was developed in the mid-20th century.
Chemical Properties[edit]
Fluroxene is chemically known as 2,2,2-trifluoroethyl vinyl ether. Its molecular formula is C4H5F3O, and it has a molecular weight of 130.08 g/mol. The presence of fluorine atoms in its structure contributes to its stability and anesthetic properties.
Pharmacology[edit]
Fluroxene acts on the central nervous system to produce a reversible loss of consciousness and sensation. Like other inhalational anesthetics, it is believed to work by modulating the activity of ion channels in the neuronal membrane, particularly those involving gamma-aminobutyric acid (GABA) and glutamate receptors.
Clinical Use[edit]
Fluroxene was used primarily in the 1960s and 1970s. It was favored for its rapid onset and recovery times compared to other anesthetics available at the time. However, its use declined due to the development of newer agents with better safety profiles and fewer side effects.
Side Effects[edit]
The use of fluroxene was associated with several side effects, including cardiovascular and respiratory depression, as well as potential hepatotoxicity. These adverse effects, along with the availability of safer alternatives, led to its discontinuation in clinical practice.
Historical Context[edit]
Fluroxene was one of the first fluorinated anesthetics to be used in clinical practice. Its development marked a significant advancement in the field of anesthesiology, paving the way for the introduction of other fluorinated anesthetics such as halothane and isoflurane.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian