Galvanic cell
== Galvanic Cell ==
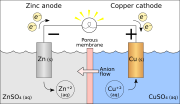
A galvanic cell, also known as a voltaic cell, is an electrochemical cell that derives electrical energy from spontaneous redox reactions taking place within the cell. It generally consists of two different metals connected by a salt bridge, or individual half-cells separated by a porous membrane.
Components[edit]
A typical galvanic cell consists of two half-cells. Each half-cell contains a metal electrode immersed in an electrolyte solution. The two half-cells are connected by a salt bridge or a porous disk that allows ions to flow between the solutions, maintaining electrical neutrality.
- **Anode**: The electrode where oxidation occurs. In a galvanic cell, the anode is the negative terminal.
- **Cathode**: The electrode where reduction occurs. In a galvanic cell, the cathode is the positive terminal.
- **Salt Bridge**: A device used to connect the oxidation and reduction half-cells of a galvanic cell. It maintains the electrical neutrality by allowing the exchange of ions.
Operation[edit]
In a galvanic cell, the oxidation-reduction reactions occur spontaneously. Electrons flow from the anode to the cathode through an external circuit, generating an electric current. The flow of electrons is driven by the difference in electrode potentials of the two half-cells.
The overall cell reaction can be split into two half-reactions:
- **Oxidation half-reaction**: Occurs at the anode.
- **Reduction half-reaction**: Occurs at the cathode.
The standard electrode potential of each half-cell can be used to calculate the cell potential of the galvanic cell using the formula: \[ E_{cell} = E_{cathode} - E_{anode} \]
Applications[edit]
Galvanic cells are widely used in various applications, including:
- **Batteries**: Many types of batteries, such as alkaline, lead-acid, and lithium-ion batteries, operate based on the principles of galvanic cells.
- **Corrosion Prevention**: Galvanic cells can be used to prevent corrosion in metals through cathodic protection.
- **Electroplating**: The process of electroplating uses galvanic cells to deposit a layer of metal onto a surface.
History[edit]
The concept of the galvanic cell was first discovered by Luigi Galvani in the late 18th century. Later, Alessandro Volta improved upon Galvani's work and created the first true battery, known as the Voltaic pile.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian