Guanidine
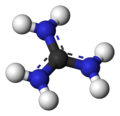
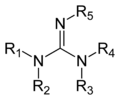
Guanidine is a compound with the formula HN=C(NH2)2. It is a colourless solid that dissolves in polar solvents. It is a strong base that is used in the production of plastics and explosives. It is found in urine as a normal product of protein metabolism. Guanidine is the compound with the highest pH value recorded.
Chemical Properties[edit]
Guanidine is a strong organic base, stronger than ammonia. It also acts as a nucleophile. The pKa of the guanidinium cation is 13.6. The related compound biguanide is also a potent base.
Uses[edit]
Guanidine is used in a variety of industrial applications, including as a plasticizer, a curing agent in epoxy resins, and a propellant for explosives. It is also used in the treatment of myasthenia gravis, a neuromuscular disease.
Health Effects[edit]
Exposure to guanidine can cause skin and eye irritation, and prolonged exposure can lead to more serious health effects such as respiratory distress and neurological damage.
See Also[edit]
Guanidine[edit]
-
Guanidinium ion 3D balls model
-
Guanidinium ion 2D skeletal structure
-
Guanidinium ion canonical forms 2D skeletal
-
Guanidine group 2D skeletal structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian