Hydrochloric acid
Hydrochloric Acid is a strong, corrosive acid that is commonly used in laboratory settings and industrial applications. It is a clear, colorless solution of hydrogen chloride (HCl) in water.
Chemical Properties[edit]
Hydrochloric acid is a strong monoprotic acid, which means it can release one proton (H+) per molecule. It has a pKa of -6.3, making it one of the strongest acids. It is also a strong electrolyte, meaning it fully dissociates into ions in solution.
Physical Properties[edit]
Hydrochloric acid is a clear, colorless to slightly yellow liquid. It has a pungent, irritating odor and is highly corrosive. It has a boiling point of 110 degrees Celsius and a melting point of -27 degrees Celsius.
Uses[edit]
Hydrochloric acid has many uses in industry and in the laboratory. It is used in the production of chlorides, fertilizers, and dyes, in electroplating, and in the photographic, textile, and rubber industries. It is also used in the laboratory for the preparation of samples for testing, in the neutralization of basic systems, and in the titration of substances.
Safety[edit]
Hydrochloric acid is a highly corrosive substance and can cause severe burns and eye damage. It is harmful if inhaled and can cause respiratory irritation. It is also harmful if swallowed. Proper safety measures should be taken when handling hydrochloric acid, including the use of personal protective equipment and proper ventilation.
See Also[edit]
-
Phase diagram of HCl and H2O
-
Hydrochloric acid 30-33%
-
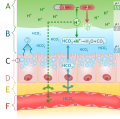
Labeled stomach mucosal layer
-
UN transport pictogram for corrosive substances
-
GHS pictogram for corrosive substances
-
GHS pictogram for harmful substances
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian