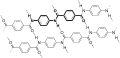
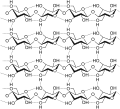
Hydrogen bond
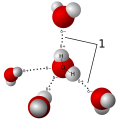
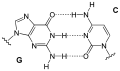
Hydrogen bond is a type of chemical bond that is formed when a hydrogen atom is attracted to an electronegative atom, such as oxygen, nitrogen, or fluorine. This bond is often denoted as H---A, where A is the electronegative atom and the dashes represent the bond.
Overview[edit]
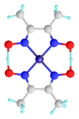
Hydrogen bonds are generally stronger than van der Waals forces, but weaker than covalent bonds or ionic bonds. They play a crucial role in many biological processes, including DNA replication and protein folding. Hydrogen bonds also contribute to the unique properties of water.
Formation[edit]
A hydrogen bond is formed when a hydrogen atom that is already part of a polar covalent bond becomes attracted to another electronegative atom. The hydrogen atom, being positively charged, is attracted to the negative charge of the electronegative atom. This forms a type of dipole-dipole attraction known as a hydrogen bond.
Properties[edit]
Hydrogen bonds have several unique properties. They are directional, meaning they have a preferred spatial arrangement. They are also relatively strong, with a typical bond energy of about 5 to 30 kJ/mol. This is significantly stronger than a van der Waals interaction, but weaker than a covalent or ionic bond.
Role in Biological Systems[edit]
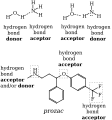
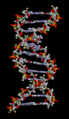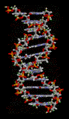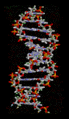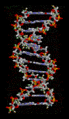
Hydrogen bonds play a crucial role in many biological systems. In DNA, hydrogen bonds between the base pairs hold the two strands of the double helix together. In proteins, hydrogen bonds contribute to the secondary and tertiary structure of the protein.
See Also[edit]
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
-
Hydrogen_bond
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian