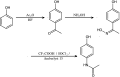
Hydroxylamine
Hydroxylamine is an inorganic compound with the formula NH2OH. The pure material is a white, unstable crystalline, hygroscopic compound. However, hydroxylamine is almost always provided and used as an aqueous solution. It is used to prepare oximes, an important functional group. It is also an intermediate in biological nitrification.
History[edit]
Hydroxylamine was first prepared as hydroxylamine hydrochloride in 1865 by the German chemist Wilhelm Clemens Lossen (1838–1906); he reacted tin with hydrochloric acid and nitric acid.
Production[edit]
Hydroxylamine is produced industrially by the raschig process, which involves the reaction of ammonia with chlorine in aqueous solution to give ammonium chloride and nitrogen. The net reaction is:
NH3 + Cl2 + H2O → NH2OH + 2 HCl
Applications[edit]
Hydroxylamine and its salts are commonly used in organic synthesis for the conversion of ketones and aldehydes to oximes, which are key intermediates in many industrial processes. It is also used in the production of pharmaceuticals, rubber, textiles, and plastics.
Safety[edit]
Hydroxylamine is a potential explosive that can detonate. It forms explosive mixtures with air and can react with oxidizing materials. It is also harmful if swallowed, inhaled or absorbed through the skin.
See also[edit]
-
Hydroxylamine
-
Beckmann Rearrangement
-
Celanese Synthesis of Paracetamol
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian