Indatuximab ravtansine
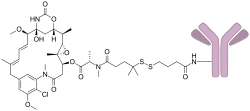
Indatuximab ravtansine is an antibody-drug conjugate (ADC) designed for the treatment of various types of cancer. It is composed of a monoclonal antibody linked to a cytotoxic agent, specifically ravtansine, which is a derivative of the potent microtubule-disrupting agent maytansine.
Mechanism of Action[edit]
Indatuximab ravtansine targets the CD138 antigen, which is commonly overexpressed on the surface of multiple myeloma cells and other hematologic malignancies. Upon binding to CD138, the ADC is internalized by the cancer cell, where the cytotoxic agent ravtansine is released. Ravtansine then binds to tubulin, disrupting the microtubule network, which is essential for cell division, ultimately leading to cell death.
Clinical Development[edit]
Indatuximab ravtansine has been evaluated in several clinical trials for its efficacy and safety in treating multiple myeloma and other cancers. Early-phase trials have shown promising results, with significant anti-tumor activity and manageable side effects.
Side Effects[edit]
Common side effects of indatuximab ravtansine include fatigue, nausea, peripheral neuropathy, and thrombocytopenia. These side effects are generally consistent with those observed with other antibody-drug conjugates.
Current Status[edit]
As of the latest updates, indatuximab ravtansine is still under investigation in clinical trials. It has not yet received approval from major regulatory agencies such as the Food and Drug Administration (FDA) or the European Medicines Agency (EMA).
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian